Aluminum melt cleaning and drossing removal flux is a chloride-fluoride salt formulation — typically based on KCl-NaCl-Na₃AlF₆ chemistry — applied to molten aluminum at 680–780°C to separate metallic aluminum trapped within surface dross, reduce dross viscosity so that entrapped metal drains back into the melt, produce a dry and easily-skimmed dross residue, and simultaneously clean the melt body of non-metallic inclusions and oxide films — with AdTech’s drossing flux product range achieving 20–45% improvement in metal recovery from dross, reducing metal loss from 55–70% trapped metal content in untreated dross down to 18–30% in flux-treated dross, representing one of the highest-return consumable investments available to secondary aluminum smelters, die casting operations, and aluminum foundries of all scales.
If your project requires the use of Aluminum Fluxes, you can contact us for a free quote.
At AdTech, we have supplied drossing and melt cleaning flux products to aluminum processing facilities across Asia, the Middle East, North America, and Europe. The economics of drossing flux treatment are straightforward and compelling once foundry operators understand what is actually inside their dross — and how much revenue they are literally skimming off and discarding.

What Is Aluminum Drossing Flux and Why Does Every Foundry Need It
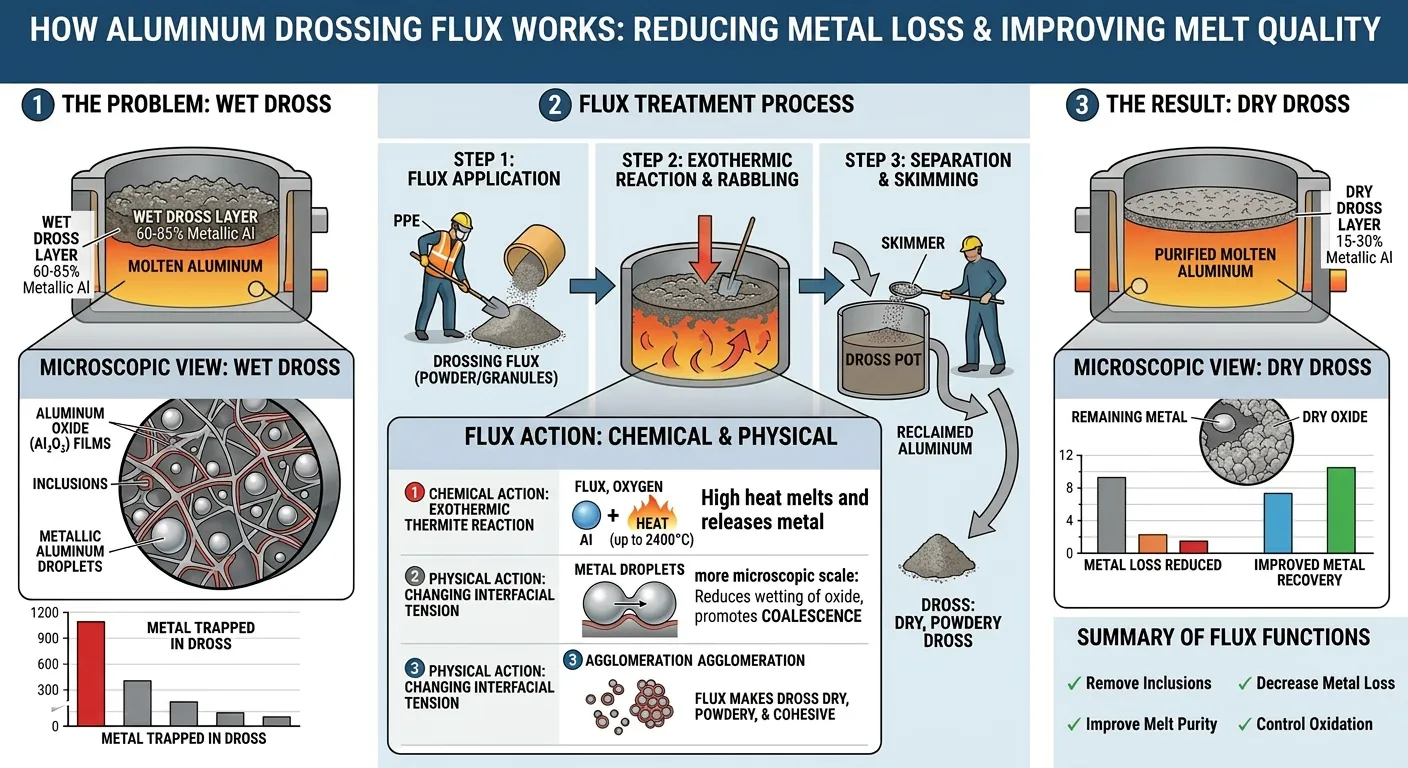
Aluminum drossing flux is a granular or powder-form inorganic salt mixture applied to the surface of molten aluminum to treat the layer of oxidized material — called dross — that accumulates on the melt surface during melting, holding, and transfer operations. The flux performs two interconnected functions: it separates metallic aluminum trapped within the dross structure (improving metal recovery and reducing material loss) and it cleans the melt surface and body of non-metallic inclusions that would otherwise be incorporated into castings.
The word “dross” covers a specific material: the surface layer that forms when molten aluminum contacts air. It is not simply waste or slag. Properly analyzed dross from an untreated secondary aluminum foundry contains 40–70% metallic aluminum — the same material the foundry paid for when purchasing the charge. The remaining 30–60% is aluminum oxide (Al₂O₃), aluminum nitride (AlN), spinel particles (MgAl₂O₄ in magnesium-bearing alloys), and flux residue from previous treatment cycles.
Without drossing flux, attempting to skim dross removes a sticky, wet material that adheres to the skimming tool and tears metal from the bath surface. The skim carries substantial metallic aluminum with it into the waste stream. With properly applied drossing flux, the dross transforms into a dry, crumbly, non-adhesive material that separates cleanly from the melt surface, leaving a bright, uncontaminated aluminum surface behind and releasing the trapped metal back into the furnace.
The Scale of the Metal Loss Problem
Consider a secondary aluminum die casting operation melting 300 tons per month:
- Typical dross generation without treatment: 4% of charge weight = 12 tons/month dross
- Metallic aluminum content in untreated dross: 55% = 6.6 tons/month metal lost
- Aluminum value at USD 2,400/ton: USD 15,840/month in trapped metal
- After drossing flux treatment, dross metal content drops to 22%: 2.64 tons lost
- Metal recovered per month through flux treatment: 3.96 tons × USD 2,400 = USD 9,504/month
- Monthly flux cost for this operation: USD 400–800
- Net monthly benefit: USD 8,700–9,100
This calculation is why every aluminum processing facility that generates significant dross volumes should treat drossing flux not as an optional consumable but as a direct-revenue product.
The Composition of Aluminum Dross: What You Are Losing Without Treatment
Understanding what dross contains physically and chemically is the foundation of making the economic case for drossing flux investment. The composition of dross varies with alloy type, melting practice, furnace atmosphere, and charge material quality, but certain patterns are consistent.
Typical Dross Composition by Aluminum Processing Type
| Process Type | Metallic Al Content | Al₂O₃ Content | AlN Content | MgO/Spinel | Other |
|---|---|---|---|---|---|
| Primary Al smelting dross | 30–50% | 35–45% | 8–15% | 1–3% | 5–10% |
| Secondary Al remelt (clean scrap) | 45–60% | 25–35% | 8–12% | 2–5% | 5–8% |
| Secondary Al (contaminated scrap) | 35–55% | 30–40% | 10–18% | 2–8% | 5–12% |
| Al-Mg alloy dross (Mg > 2%) | 40–60% | 20–30% | 5–10% | 15–25% | 3–8% |
| Die casting holding furnace dross | 50–65% | 20–32% | 5–10% | 2–6% | 5–10% |
| Untreated scrap remelting dross | 30–50% | 30–45% | 12–20% | 2–8% | 5–12% |
Why Metallic Aluminum Becomes Trapped in Dross
The mechanism of metal entrapment in dross is physical rather than chemical. When the oxide skin on a molten aluminum surface is disturbed — by turbulence during charging, stirring, metal transfer, or the deliberate action of skimming — the oxide skin ruptures and folds. Where the oxide skin folds over itself, it encloses pockets of liquid aluminum. These enclosed metal droplets are surrounded by oxide that acts as a barrier preventing them from returning to the bulk metal by surface tension forces.
The oxide-enclosed metal droplets range in size from sub-millimeter to several millimeters. As dross accumulates and cools slightly at the surface, these droplets become increasingly immobile. The dross structure becomes a solid or semi-solid matrix of oxide with liquid metal droplets distributed throughout — essentially a sponge of alumina with liquid metal filling the pores.
Drossing flux reduces the viscosity and surface tension of this oxide matrix, allowing the pores to collapse and releasing the trapped metal droplets to coalesce and drain back through the now-more-fluid dross structure into the bulk melt.
How Drossing Removal Flux Works: Chemistry and Metallurgical Mechanisms
Mechanism 1: Viscosity Reduction of the Oxide Matrix
The primary mechanism of drossing flux action is dissolution of aluminum oxide (Al₂O₃) components within the dross matrix by fluoride ions from the flux. Specifically, cryolite (Na₃AlF₆) and potassium fluoride (KF) components of the flux react with Al₂O₃ to form soluble aluminate-fluoride complexes.
This dissolution process reduces the melting point of the oxide matrix from above 2000°C (pure Al₂O₃ melts at 2072°C) to 700–800°C in the presence of fluoride flux — meaning the oxide phase becomes semi-liquid at aluminum processing temperatures rather than remaining a rigid solid. The liquid oxide phase has much lower viscosity and allows the enclosed aluminum droplets to drain freely.
The practical visual result: before flux application, the dross is gray, heavy, and sticky. After 3–5 minutes of flux contact, it becomes pale, light, and crumbly — experienced operators describe this as the dross “breathing” or “opening up” as the metallic aluminum drains back through the treated structure.
Mechanism 2: Interfacial Tension Modification
The second mechanism operates at the aluminum-oxide interface. Chloride salt components (KCl, NaCl) of the flux reduce the interfacial surface tension between metallic aluminum droplets and the surrounding aluminum oxide matrix. Lower interfacial tension allows small metal droplets to coalesce more easily — small droplets merge into larger droplets that have sufficient weight to overcome the surface tension barrier and drain through the oxide matrix back into the bulk melt.
This explains why the metal recovery improvement from drossing flux is greater for fine, dispersed metal droplets (which have the highest surface tension resistance to drainage) than for large metal inclusions that would drain under gravity without flux assistance.
Mechanism 3: Carrier Salt Penetration
The KCl-NaCl carrier salts in drossing flux melt at approximately 657°C (at the eutectic composition) and flow as a low-viscosity liquid across and through the dross structure. This liquid carrier phase carries the active fluoride components into the interior of the dross mass, where they can contact and react with oxide phases throughout the dross thickness — not just at the surface.
This penetration mechanism is why application technique matters: simply sprinkling flux on top of the dross and skimming immediately delivers minimal benefit because the flux has not had time to penetrate into the dross interior where the majority of trapped metal resides. Working the flux into the dross body with a perforated skimmer, and allowing adequate contact time (3–5 minutes minimum), is essential for complete treatment.
Mechanism 4: Aluminum Nitride (AlN) Stabilization
Aluminum nitride in dross presents a specific challenge: AlN reacts exothermically with moisture to generate ammonia (NH₃) gas. This reaction can cause dross to “burn” when exposed to humid air — a safety concern and a source of toxic gas emission. Some drossing flux formulations include components that stabilize aluminum nitride by converting it to less-reactive compounds, reducing the burning tendency of treated dross.
AdTech’s heavy-duty drossing flux includes AlN stabilization chemistry, making it the correct choice for secondary aluminum operations handling highly contaminated scrap where AlN content in dross is elevated.
Types of Aluminum Melt Cleaning Flux: Drossing, Refining, and Cleaning Functions
Understanding the Distinctions Between Melt Cleaning Flux Types
The term “melt cleaning flux” covers several functionally distinct products that are sometimes confused with each other. Understanding the distinction ensures the correct product is specified for each function.
| Flux Category | Primary Target | Chemistry Basis | Application Zone | Visual Result |
|---|---|---|---|---|
| Drossing flux | Surface dross metal recovery | KCl-NaCl-Na₃AlF₆-KF | Dross surface layer | Dry, crumbly, non-sticky dross |
| Degassing flux | Dissolved hydrogen removal | KCl-NaCl-Na₃AlF₆-K₂TiF₆ | Melt body (injection) | Bubble release; foam on surface |
| Refining / inclusion flux | Fine bifilm removal | High-fluoride KCl-NaCl | Melt body (injection) | Cleaner melt surface; less gray |
| Furnace wall cleaning flux | Sintered oxide wall buildup | High-fluoride Na₃AlF₆-KF | Furnace walls and hearth | Oxide dissolution from refractory |
| Covering flux | Melt surface protection | KCl-NaCl base (low fluoride) | Melt surface blanket | Protective salt layer |
When to Use Each Type
Use drossing flux when:
- Dross buildup on the melt surface is the primary issue.
- Metal loss from dross is measurable and economically significant.
- Dross is wet, sticky, and difficult to skim cleanly.
- Post-skim melt surface appears dull or gray.
Use refining flux when:
- Casting inclusions and mechanical property scatter are the primary issues.
- Surface dross is manageable but internal casting quality is poor.
- Alkali metal contamination (Na, Ca) from scrap is suspected.
- Working with alloys sensitive to bifilm inclusions (A356, A357 for automotive).
Use furnace wall cleaning flux when:
- Furnace capacity has reduced from wall buildup over time.
- Wall-attached oxide is breaking off and entering the melt.
- Furnace efficiency has dropped without other explanation.
- During planned maintenance shutdowns.
The most common operational error we observe is using drossing flux as a general “melt cleaner” when the real quality problem is dissolved hydrogen — a function that drossing flux cannot address. Always match flux type to the specific metallurgical problem.

Technical Specifications for Drossing Flux Products
Chemical Composition Requirements
| Parameter | Standard Drossing Flux | Heavy-Duty Drossing Flux | Low-Salt Drossing Flux |
|---|---|---|---|
| KCl content | 50–62% | 45–55% | 15–30% |
| NaCl content | 18–27% | 15–23% | 10–18% |
| Na₃AlF₆ (cryolite) | 12–18% | 14–22% | 8–14% |
| KF content | 5–12% | 10–18% | 4–10% |
| AlN stabilizer | Not included | 2–5% | Optional |
| Moisture content | ≤ 0.30% | ≤ 0.25% | ≤ 0.35% |
| Total chlorides | 55–72% | 48–65% | 20–40% |
| Total fluorides | 12–22% | 18–32% | 10–20% |
Physical Properties
| Property | Standard Grade | Heavy-Duty Grade | Test Method |
|---|---|---|---|
| Physical form | Fine powder | Granular or powder | Visual |
| Particle size | 0.1–0.5mm | 0.3–2.0mm | Sieve analysis |
| Bulk density | 0.90–1.15 g/cm³ | 0.95–1.20 g/cm³ | Cylinder method |
| Melting point | 650–700°C | 640–690°C | DSC analysis |
| Application temp | 700–760°C | 700–780°C | Thermocouple |
| pH (10% solution) | 7.5–9.5 | 7.5–9.5 | pH meter |
| Shelf life (sealed) | 24 months | 24 months | Manufacturing date |
| Packaging | 25kg sealed bags | 25kg sealed bags | Moisture-proof |
Performance Specifications
| Performance Parameter | Untreated Dross Baseline | Standard Flux Treatment | Heavy-Duty Treatment |
|---|---|---|---|
| Metallic Al in dross (%) | 50–65% | 28–40% | 18–28% |
| Dross density | High (heavy, wet) | Medium | Low (light, dry) |
| Metal recovery improvement | Baseline | +15–28% | +25–42% |
| Skimmability | Poor (sticky) | Good | Excellent |
| Post-skim surface appearance | Dull, gray | Mostly bright | Bright, clean |
| Treatment time required | N/A | 3–6 min/m² dross | 4–8 min/m² dross |
| Flux dosing rate | N/A | 5–12 kg/ton dross | 8–18 kg/ton dross |
Metal Recovery Calculations: The Economic Case for Drossing Flux
Calculating Your Operation’s Dross Metal Loss
This calculation framework applies to any aluminum processing operation and allows precise quantification of the financial benefit from drossing flux investment.
Step 1: Determine monthly dross generation volume
Dross generation rate (%) × Monthly aluminum charge weight (tons) = Monthly dross weight (tons)
Step 2: Determine current metallic aluminum content in dross
Sample dross regularly using acid dissolution or hydrogen release method to measure metallic Al%. If no measurement exists, use 55% as a conservative estimate for secondary aluminum operations.
Step 3: Calculate current monthly metal loss value
Monthly dross weight × Metallic Al% × Current aluminum price (USD/ton) = Monthly metal loss value (USD)
Step 4: Estimate post-flux treatment metallic Al% in dross
Using AdTech heavy-duty drossing flux: target 20–25% metallic Al in treated dross.
Using AdTech standard drossing flux: target 28–35% metallic Al in treated dross.
Step 5: Calculate monthly metal recovery improvement value
(Untreated Al% − Treated Al%) × Monthly dross weight × Aluminum price = Monthly recovery value
Step 6: Calculate monthly flux cost
Flux dosing rate (kg/ton dross) × Monthly dross weight (tons) × Flux price (USD/kg)
Step 7: Calculate net monthly benefit
Monthly recovery value − Monthly flux cost = Net monthly benefit.
Sample Calculation for a Medium-Scale Die Casting Operation
| Parameter | Value |
|---|---|
| Monthly aluminum charge | 250 tons |
| Dross generation rate | 3.5% |
| Monthly dross weight | 8.75 tons |
| Current metallic Al in dross | 58% |
| Monthly metal loss (untreated) | 5.075 tons |
| Aluminum value | USD 2,450/ton |
| Monthly loss value | USD 12,434 |
| Post-treatment metallic Al (AdTech HD flux) | 22% |
| Monthly metal loss (treated) | 1.925 tons |
| Monthly metal recovered | 3.15 tons |
| Monthly recovery value | USD 7,718 |
| Flux consumption (10 kg/ton dross) | 87.5 kg |
| Flux cost (USD 5.50/kg) | USD 481 |
| Net monthly benefit | USD 7,237 |
| Annual net benefit | USD 86,844 |
This calculation demonstrates why drossing flux represents one of the most favorable cost-to-benefit ratios of any consumable in aluminum processing.
Correct Application Procedure: Step-by-Step Dross Treatment
Pre-Treatment Preparation
The success of drossing flux treatment depends heavily on preparation steps that many operators skip:
Temperature verification: Melt temperature must be within the 700–760°C range before applying drossing flux. Below 680°C, the flux melting point approaches melt temperature, reducing fluidity and penetration into the dross. Above 780°C, accelerated surface oxidation generates new dross faster than treatment can handle it.
Allow dross to accumulate: Premature skimming of thin, scattered dross reduces the economics of flux treatment. Allow dross to accumulate to a thickness where flux application is economically meaningful — typically when the dross layer covers more than 60–70% of the melt surface.
Gather equipment: Perforated steel skimmer (holes allow metal to drain while skimming oxide), dross cart or container positioned beside the furnace, weighed flux dose ready for application, and appropriate PPE.
Step-by-Step Application Sequence
Step 1: Reduce melt agitation
Stop any stirring, charging, or metal movement 2–3 minutes before applying drossing flux. Allow the melt surface to calm and the dross layer to consolidate.
Step 2: Apply flux uniformly across dross surface
Spread the weighed flux dose uniformly across the entire dross surface — not just the center or edges. Use a spreading motion that covers all visible dross. Avoid dumping the entire flux dose in one location.
Step 3: Work flux into the dross body
This is the most critical and most commonly skipped step. Using a perforated skimmer, work the flux into the dross by pressing it gently downward and making folding motions that bring the flux-dross mixture into contact throughout the dross thickness. The flux must reach the interior of the dross mass where the majority of trapped metal resides.
Step 4: Allow contact time
After initial working, allow the treated dross to sit undisturbed for 3–5 minutes. During this period, the flux melts, penetrates the oxide matrix, reduces its viscosity, and allows metallic aluminum droplets to drain downward back through the dross into the melt.
Step 5: Visual assessment
Treated dross changes appearance noticeably — the color lightens, the surface becomes less shiny (indicating less surface metal), and the texture becomes drier and more granular. If the dross still appears wet and shiny after contact time, apply a supplementary dose of flux (25–50% of original dose) and allow additional contact time.
Step 6: Skim with perforated tool
Using the perforated skimmer, push the treated dross to one side of the furnace in a smooth, deliberate motion. Avoid repeated back-and-forth scraping that re-incorporates flux-treated dross back into the melt. The perforated skimmer allows the last traces of liquid metal to drain back through the tool as the dross is removed.
Step 7: Verify melt surface condition
After skimming, inspect the melt surface. A bright, reflective surface indicates successful dross removal. Remaining dark or gray areas indicate incomplete dross removal — repeat flux application to these areas specifically.
Step 8: Post-treatment considerations
The skimmed dross should be dry, light, and non-sticky. Transfer it directly to a dross processing container. If a mechanical dross press is available, process immediately while still warm to recover additional metallic aluminum.
Drossing Flux for Different Aluminum Alloys and Furnace Types
Alloy-Specific Drossing Requirements
| Alloy Family | Dross Generation Rate | Recommended Flux Type | Special Consideration |
|---|---|---|---|
| A356 / A357 (Al-Si-Mg) | Medium-High (3–5%) | Standard + HD alternating | Mg spinel in dross; higher fluoride needed |
| A380 / ADC12 (Al-Si-Cu) | Medium (2.5–4%) | Standard drossing flux | Copper inclusions; standard treatment effective |
| 5xxx (Al-Mg, >3% Mg) | Very High (5–10%) | Heavy-duty with AlN stabilizer | Mg dramatically increases dross rate |
| 1xxx high-purity Al | Low (1.5–2.5%) | Standard drossing flux (low dose) | Very clean; standard treatment sufficient |
| Al-Cu (2xx alloys) | Medium (2.5–4%) | Standard drossing flux | Cu inclusions; standard chemistry adequate |
| Secondary mixed scrap | Very High (4–8%) | Heavy-duty drossing flux | Most demanding; high AlN content in dross |
| Brass / Al-Zn alloys | High (3–6%) | Heavy-duty drossing flux | Zinc volatility; ventilation critical |
Furnace Type Application Adjustments
| Furnace Type | Dross Characteristics | Flux Application Method | Dose Adjustment |
|---|---|---|---|
| Reverberatory (gas-fired) | High volume; moisture from combustion | Standard procedure | Standard dose |
| Electric induction furnace | Moderate volume; cleaner | Reduced contact time needed | -15% from standard |
| Electric resistance holding | Low volume; clean | Minimal treatment needed | -25% from standard |
| Crucible (small batch) | Variable; high turnover | Manual application | Per-batch calculation |
| Rotary furnace (secondary Al) | Very high volume; heavy dross | Mechanical assist recommended | +20% from standard |
| Tilting melting furnace | Variable by operation | Standard procedure | Standard dose |
Secondary Aluminum Smelter Specific Guidance
Secondary aluminum smelters face the most demanding drossing flux requirements in the industry. Highly contaminated scrap (painted, coated, oily) generates dross with:
- Higher organic contamination that increases dross volume.
- Higher AlN content from nitrogen atmosphere contact.
- Higher oxide loading from surface contamination oxidation.
- Greater variability in dross composition between batches.
For secondary smelter operations, we recommend AdTech DR-2 heavy-duty drossing flux at 12–18 kg per ton of dross, combined with mechanical dross pressing equipment for maximum metal recovery. The combination of flux treatment plus mechanical pressing achieves metallic aluminum content in final dross residue of 12–18% — approaching the theoretical minimum for practical operations.
Combining Drossing Flux with Degassing Treatment and Ceramic Foam Filtration
The Complete Aluminum Melt Treatment Sequence
Drossing flux treatment is most effective when integrated into a complete melt treatment program rather than used in isolation. The correct treatment sequence:
| Step | Treatment | Product | Purpose |
|---|---|---|---|
| 1 | Initial charge melt and temperature adjustment | N/A | Reach treatment temperature |
| 2 | Drossing flux treatment | AdTech DR-1 or DR-2 | Remove accumulated dross, recover metal |
| 3 | Skim treated dross | N/A | Remove oxide and flux residue |
| 4 | Degassing treatment | AdTech DG-1 with rotary unit | Remove dissolved hydrogen |
| 5 | Post-degassing dross removal | AdTech DR-1 | Remove byproduct dross from degassing |
| 6 | Covering flux application | AdTech CV-1 | Protect treated melt until casting |
| 7 | Grain refiner addition | AlTi5B1 rod | Grain refinement (if required) |
| 8 | Transfer to casting station | N/A | Minimize reoxidation during transfer |
| 9 | Ceramic foam filtration | AdTech Al₂O₃ 30–40 PPI | Capture residual fine inclusions |
| 10 | Casting | N/A | Pour into mould through filter |
Why Sequence Matters
Attempting degassing treatment before drossing is a common operational error. Existing dross on the melt surface during degassing:
- Insulates the near-surface metal zone from hydrogen bubble collection.
- Absorbs degassing flux preferentially before it can act on dissolved hydrogen in the melt body.
- Generates additional inclusions when degassing bubbles break through the dross layer.
- Reduces overall degassing efficiency by 20–35% compared to treating a clean melt surface.
Always skim dross before beginning degassing treatment.
Why Drossing Flux and Ceramic Foam Filtration Are Complementary
Drossing flux addresses the coarse, surface-level aluminum oxide and dross problem. Ceramic foam filtration (using AdTech Al₂O₃ 30–40 PPI filters in the gating system) addresses the fine inclusion population that remains after dross skimming — sub-millimeter oxide bifilms, spinel particles, and fine intermetallic particles that are too light to be skimmed and too small to be removed by drossing flux alone.
The two technologies target different inclusion size ranges and cannot effectively substitute for each other. A properly treated and filtered aluminum melt achieves casting quality that neither process achieves independently.
Safety, Storage, and Environmental Compliance
Critical Safety Considerations
Moisture hazard: This is the most serious safety concern with drossing flux. Chloride-fluoride flux materials absorb atmospheric moisture aggressively. If moisture-contaminated flux contacts 700–760°C molten aluminum, the moisture vaporizes instantaneously, generating violent steam that can spray molten metal. Always verify container integrity before use. Never introduce damp or caking flux into a molten aluminum bath.
HCl and HF gas generation: During flux treatment, hydrogen chloride (HCl) and hydrogen fluoride (HF) gases are generated as reaction byproducts. Both are respiratory irritants and corrosives. All flux treatment must be conducted with adequate local exhaust ventilation operating. OSHA PEL for HCl is 5 ppm ceiling; for HF is 3 ppm TWA.
Thermal hazards: Drossing flux materials melt at 650–700°C and behave as energetic molten liquids during treatment. Contact with bare skin causes severe thermal and chemical burns. Full PPE is mandatory.
AlN reaction with moisture: Dross containing aluminum nitride (AlN) reacts with moisture in the air to generate ammonia (NH₃) and potentially hydrogen gas. Do not store freshly skimmed dross in sealed containers — allow it to cool in open dross carts with ventilation.
Required PPE for Dross Treatment Operations
| Task | Minimum PPE Required |
|---|---|
| Flux bag handling and weighing | Safety glasses, N95 respirator, nitrile gloves |
| Flux application to molten dross | Full face shield, P100 respirator, heat-resistant gloves, FR clothing |
| Dross skimming with perforated tool | Full face shield, P100 respirator, heat-resistant gloves, FR clothing |
| Dross transfer to press or cart | Face shield, P100 respirator, heat-resistant gloves |
| Mechanical dross pressing | Full face shield, heat-resistant full coverage, P100 respirator |
Storage Requirements
| Storage Parameter | Requirement |
|---|---|
| Container condition | Sealed, moisture-proof (original packaging) |
| Relative humidity | Below 60% in storage area |
| Temperature range | 5–35°C ambient |
| Floor condition | Elevated on pallets; no floor contact |
| Proximity to water | Store away from water sources, drains, rain exposure |
| Stack height | Maximum 3 pallet layers; follow packaging label |
| Shelf life | 24 months from manufacture date in original sealed packaging |
| Opened containers | Reseal immediately; use within 48 hours of opening |
Environmental Disposal of Spent Dross
Treated dross (salt slag) after drossing flux treatment contains residual chloride and fluoride salts from the flux, aluminum oxide, and aluminum nitride. In most regulatory jurisdictions:
- Aluminum salt slag is classified as hazardous waste due to AlN water-reactivity (generates NH₃ and potentially H₂ gas on water contact)
- Disposal requires licensed hazardous waste contractors with appropriate manifesting.
- Some jurisdictions permit dedicated aluminum salt slag recycling facilities that recover the salt component and process the residual oxide for non-metallic applications.
AdTech provides Material Safety Data Sheets (MSDS/SDS) for all flux products that include waste disposal classification information applicable to major regulatory frameworks (RCRA in the USA, EU REACH, and equivalent national regulations).
AdTech Drossing Flux Product Range and Ordering Information
Complete Product Specifications
| Product | Type | Best Application | Dosing Rate | Package Size |
|---|---|---|---|---|
| AdTech DR-1 | Standard drossing flux | Die casting, foundry gravity casting | 5–12 kg/ton dross | 25kg sealed bag |
| AdTech DR-2 | Heavy-duty drossing flux | Secondary Al smelting; high-Mg alloys | 8–18 kg/ton dross | 25kg sealed bag |
| AdTech DR-3 | Low-chloride drossing flux | EU-regulated markets; emission limits | 6–14 kg/ton dross | 25kg sealed bag |
| AdTech MP-1 | Multipurpose (drossing + degassing) | Small foundries; simplified treatment | 2–4 kg/ton Al | 25kg sealed bag |
| AdTech CL-1 | Furnace wall cleaning flux | Maintenance shutdowns; wall buildup | 10–20 kg/m² oxide | 25kg sealed bag |
Integration with AdTech Complete Flux Program
AdTech’s drossing flux products integrate with our complete aluminum melt treatment system:
- AdTech DG-1 / DG-2: Degassing flux for hydrogen removal via rotary unit or lance injection.
- AdTech DR-1 / DR-2: Drossing flux for surface dross treatment and metal recovery.
- AdTech CV-1: Covering flux for melt surface protection during holding.
- AdTech RF-1: Refining flux for fine inclusion and alkali metal removal.
All AdTech flux products are manufactured under ISO 9001:2015 quality management certification, supplied with lot-specific chemical analysis certificates, and available with Safety Data Sheets in required languages.
Minimum Order and Lead Time
Standard minimum order: 10 bags (250kg) per product grade. Pallet quantities (1,000kg / 40 bags) qualify for volume pricing. Standard lead time from order confirmation: 7–15 business days for stocked formulations. Custom formulations or low-chloride grades: 15–25 business days.
Frequently Asked Questions (FAQs)
Q1: What is aluminum drossing flux and what does it do?
Aluminum drossing flux is a chloride-fluoride salt mixture — typically containing potassium chloride (KCl), sodium chloride (NaCl), cryolite (Na₃AlF₆), and potassium fluoride (KF) — applied to the dross layer on molten aluminum to separate trapped metallic aluminum from the oxide matrix, reduce dross viscosity so that enclosed metal droplets coalesce and drain back into the melt, and produce a dry, easily-skimmed dross residue. Without flux treatment, dross contains 40–70% metallic aluminum that is discarded as waste. With proper drossing flux application, this trapped metal is recovered into the melt, reducing material loss and improving overall process yield by 15–45% depending on baseline dross quality and treatment method.
Q2: How much drossing flux should I use per ton of aluminum?
Drossing flux dosing is typically calculated per ton of dross being treated, not per ton of aluminum in the furnace, because the flux acts on the dross layer specifically. Standard drossing flux: 5–12 kg per ton of dross. Heavy-duty drossing flux for secondary aluminum or high-magnesium alloys: 8–18 kg per ton of dross. As a rough practical guide, if your operation generates approximately 3% dross by weight, you need approximately 0.15–0.5 kg of drossing flux per ton of aluminum charged. Under-dosing is the most common application error — always dose to specification because the cost of flux is a small fraction of the metal recovered.
Q3: What is the difference between drossing flux and degassing flux for aluminum?
Drossing flux and degassing flux address completely different metallurgical problems. Drossing flux acts on the surface dross layer to separate trapped metallic aluminum from oxide, reducing metal loss and improving melt surface cleanliness. It does not remove dissolved hydrogen from the melt body. Degassing flux (also called refining flux in some contexts) is injected into the melt body via lance or rotary degassing unit to generate fine bubbles that carry dissolved hydrogen to the surface and remove it — reducing casting porosity. Both are necessary for comprehensive melt treatment: dross removal before degassing produces the best combined results. Using drossing flux to try to address hydrogen porosity problems will not work, and vice versa.
Q4: How do I know if my drossing flux treatment is working correctly?
Several indicators confirm effective drossing flux treatment: (1) Visual change in dross appearance — treated dross becomes pale, dry, and granular versus the gray, wet, sticky appearance of untreated dross; (2) Reduced dross mass — effectively treated dross is significantly lighter per unit volume than untreated dross because the metal has drained out; (3) Clean melt surface after skimming — a bright, reflective aluminum surface indicates thorough dross removal; (4) Measurable improvement in metal yield — track the weight of skimmed dross before and after implementing flux treatment; if metallic aluminum content in dross drops from 55% to 22%, the weight of material being discarded as dross should drop proportionally; (5) Reduced casting inclusions from surface dross sources — fewer surface-related inclusion defects in castings produced after proper drossing treatment.
Q5: Can I make my own aluminum drossing flux from salt and other materials?
Technically, potassium chloride and sodium chloride alone provide some of the carrier function of commercial drossing flux, but they lack the fluoride components (cryolite, KF) that provide the critical oxide dissolution and viscosity reduction mechanisms. Without fluoride chemistry, the chloride salts wet the dross surface but cannot penetrate and dissolve the alumina matrix — the fundamental mechanism that releases trapped metal. Additionally, formulating flux chemistry in-house with consistent moisture content below the critical 0.30% threshold requires controlled processing and testing that is impractical outside a dedicated chemical manufacturing environment. Moisture in homemade flux creates explosion risk when it contacts molten aluminum. Commercial drossing flux from a qualified supplier provides consistent chemistry, controlled moisture, and specific technical support — the economics of metal recovery make the small per-kilogram cost of commercial flux trivially justified.
Q6: What happens if I apply drossing flux to aluminum that is too cold?
Applying drossing flux to aluminum at temperatures below approximately 680°C produces poor results for a specific chemical reason: the flux melting point (approximately 650–680°C at eutectic composition) approaches the melt temperature, leaving the flux as a viscous, semi-solid material rather than the freely-flowing liquid needed to penetrate dross structure. The flux may sit on top of the dross without distributing into the oxide matrix interior. The practical result is minimal metal recovery improvement and wasted flux. Always verify melt temperature is within 700–760°C before beginning drossing flux treatment. If the furnace has cooled below target range, allow it to return to temperature before applying flux.
Q7: How often should I apply drossing flux in an aluminum die casting operation?
Application frequency depends on dross generation rate, which varies with alloy type, scrap quality, furnace atmosphere, and metal transfer turbulence. Most die casting holding furnace operations benefit from drossing flux treatment every 4–8 hours of continuous operation or whenever dross accumulation covers more than 50–60% of the melt surface. In secondary aluminum operations with contaminated scrap, more frequent treatment (every 2–4 hours) may be necessary. The economic signal for increasing treatment frequency: when the weight of discarded dross per ton of aluminum charged exceeds 3.5–4%, the dross is likely accumulating faster than the current treatment schedule removes it. Track dross weight systematically and use it as the primary process control indicator for treatment frequency decisions.
Q8: Does drossing flux affect the composition of the aluminum alloy?
When applied and used correctly, drossing flux does not measurably alter aluminum alloy composition. The flux components (KCl, NaCl, Na₃AlF₆, KF) do not dissolve into the aluminum melt in significant quantities at normal treatment temperatures and contact times. The small amounts of sodium and fluoride that may contact the melt surface are in equilibrium with sodium and fluoride already present in the oxide layer, not dissolving into the metal body. However, two specific risks exist: (1) If fluoride-containing flux is in prolonged contact with high-magnesium aluminum alloys, trace fluoride pickup is possible — monitor Mg content after implementing heavy-duty flux treatment; (2) Incomplete skimming that leaves flux residue on the melt surface can incorporate trace chloride into the metal, appearing as porosity in castings. Always skim thoroughly after drossing flux treatment and verify the melt surface is clean before casting.
Q9: What is the best way to dispose of dross after treating it with aluminum drossing flux?
Dross after flux treatment (called salt slag or black dross in the secondary aluminum industry) contains residual chloride and fluoride salts from the flux, aluminum oxide, and aluminum nitride. In most jurisdictions this material is classified as hazardous waste due to the water-reactive nature of AlN, which generates ammonia gas on contact with moisture. Proper disposal requires: (1) cooling the hot dross in a ventilated area before containment — never seal hot dross containing AlN in closed containers; (2) engaging a licensed hazardous waste contractor for removal and disposal with appropriate waste manifest documentation; (3) investigating whether a dedicated aluminum salt slag recycler operates in your region — these facilities process salt slag to recover the chloride salt (which they return to flux manufacturers for reformulation) and process the aluminum oxide residue for other industrial uses, achieving near-zero waste processing. Contact AdTech’s technical team for regional salt slag recycling facility referrals where available.
Q10: How does aluminum drossing flux interact with ceramic foam filtration?
Drossing flux treatment and ceramic foam filtration target different inclusion populations and work synergistically. Drossing flux removes coarse, surface-level dross (primarily oxide films and oxide clusters visible to the naked eye) before the metal reaches the gating system. Ceramic foam filtration (using AdTech Al₂O₃ 30–40 PPI filters placed in the gating system) captures the fine oxide bifilm population — sub-millimeter inclusions that are too light to be skimmed by any surface treatment and too small to be removed by drossing flux alone. These fine bifilms are the primary cause of elongation shortfalls, fatigue life reduction, and machined surface porosity in aluminum castings. The correct sequence: complete drossing flux treatment and skimming in the furnace, then filter the metal through an Al₂O₃ ceramic foam filter during mould filling. Foundries that implement both processes consistently achieve lower casting rejection rates than those using either process alone.
Summary: Implementing an Effective Aluminum Dross Treatment Program
Aluminum melt cleaning and drossing removal flux is one of the highest-return consumable investments in aluminum processing. The economics are compelling and straightforward: dross contains valuable metallic aluminum that conventional skimming without flux treatment discards as waste. Proper drossing flux treatment recovers 25–45% more metal from dross, directly improving aluminum yield and reducing material cost.
The key principles for an effective dross treatment program:
Match flux to alloy: High-magnesium alloys (A356, A357, 5xxx series) and secondary aluminum operations with contaminated scrap require heavy-duty drossing flux with elevated fluoride content and AlN stabilization. Standard drossing flux is appropriate for primary aluminum and low-Mg alloys.
Application technique determines results: Flux must penetrate into the dross body interior — surface application without working the flux into the dross structure recovers only a fraction of the available metal. Allow minimum 3–5 minutes contact time after working flux into dross.
Integrate into complete melt treatment: Dross before degassing, degassing before casting, filtration during mould filling. The sequence matters and each step builds on the previous one.
Measure results systematically: Track dross weight before and after treatment, estimate metallic aluminum content, and calculate actual metal recovery improvement. Without measurement, there is no systematic basis for optimizing the treatment program.
Never compromise on moisture content: Wet or moisture-contaminated drossing flux creates serious safety hazards and delivers poor metallurgical performance. Proper storage in sealed, dry conditions is non-negotiable.
AdTech’s drossing flux product range — including standard DR-1, heavy-duty DR-2, and low-chloride DR-3 formulations — provides the complete solution for aluminum melt dross management across all foundry and smelting applications, manufactured under ISO 9001:2015 quality management with full chemical analysis certification and technical application support.
This article is prepared by AdTech’s technical editorial team. Product specifications, dosing guidelines, and performance data reflect current AdTech formulations as of 2025–2026. Contact AdTech’s technical sales team for application-specific recommendations, sample requests, and current pricing.

