The fluxes used for aluminum are salt-based chemical compounds — primarily chloride and fluoride salts — applied to molten aluminum to remove oxide inclusions, reduce dross formation, extract dissolved hydrogen, and remove alkali metal impurities. The most common aluminum fluxes include mixtures of sodium chloride (NaCl) and potassium chloride (KCl) as the base, combined with reactive fluoride compounds such as cryolite (Na₃AlF₆), calcium fluoride (CaF₂), or aluminum fluoride (AlF₃) depending on the specific treatment objective. At AdTech, we formulate and supply aluminum fluxes to casthouse operations, secondary smelters, and die casting foundries, and our consistent field experience confirms that no single flux serves all purposes — the correct flux selection depends on whether you are covering the melt surface, removing inclusions, extracting alkali metals, or refining dross.
If your project requires the use of Aluminum Flux, you can contact us for a free quote.
Why Does Aluminum Need Flux Treatment?
Aluminum’s chemical reactivity is both its most useful property and its biggest processing challenge. At melt temperatures of 680–780°C, liquid aluminum oxidizes almost instantaneously when it contacts atmospheric oxygen, forming a solid aluminum oxide (Al₂O₃) skin on the melt surface. This oxide layer, if continuously disrupted by melt turbulence, generates oxide inclusions that remain suspended throughout the metal volume and become trapped in solidified castings, causing defects.
Beyond surface oxidation, aluminum melts — particularly those prepared from recycled scrap — absorb dissolved hydrogen from moisture in the charge material and from atmospheric humidity, contain alkali metal impurities (sodium, calcium, potassium) from various sources, and carry non-metallic inclusions from refractory erosion, alloying additions, and improper handling.
Fluxes address these problems through chemical reactions, physical wetting, and density separation. Understanding which problem each flux type targets is essential for effective melt treatment.

The Core Problems That Aluminum Flux Treats
| Problem | Source | Consequence if Untreated | Flux Treatment |
|---|---|---|---|
| Surface oxidation | O₂ contact at melt surface | Oxide film entrapment, dross loss | Covering flux |
| Oxide inclusion suspension | Melt turbulence, bifilm formation | Porosity, mechanical property reduction | Cleaning/refining flux |
| Dross formation | Surface oxide accumulation | Metal loss in dross, thermal inefficiency | Drossing flux |
| Dissolved hydrogen | Moisture, scrap contamination | Gas porosity in castings | Degassing flux (or rotary degassing) |
| Sodium contamination | Salt flux residues, some scrap sources | Embrittlement, surface blistering | Alkali removal flux |
| Calcium contamination | Master alloys, some scrap | Grain structure modification, cracking | Alkali removal flux |
| Magnesium depletion (in some alloys) | Oxidation during holding | Off-spec composition | Protective covering flux |
What Are the Main Types of Flux Used for Aluminum?
Aluminum fluxes are categorized by their primary function rather than their specific chemical composition, because multiple chemical systems can achieve similar functional results. The major categories recognized in industrial practice are:
Covering fluxes: Applied to the melt surface to form a protective liquid or semi-liquid blanket that prevents atmospheric oxygen and moisture from contacting the aluminum. These are the most widely used flux type in foundry practice and are applied during the entire holding period between melting and casting.
Drossing fluxes: Applied specifically to the dross layer that accumulates on the melt surface. Their function is to lower the surface tension and wetting angle between the oxide particles and residual metallic aluminum trapped in the dross, causing the metal to coalesce and drain back into the melt, increasing metal recovery from dross and reducing dross disposal volume.
Cleaning and refining fluxes: Formulated to penetrate the melt and react with suspended inclusions, promoting their flotation to the melt surface where they can be skimmed. These fluxes reduce the bulk inclusion content of the melt and improve filtration effectiveness by agglomerating fine particles into larger clusters.
Degassing fluxes: Reactive compounds (historically hexachloroethane, now largely replaced by gas injection) that generate reactive gases within the melt. These gases bubble through the metal, collecting dissolved hydrogen and carrying it to the surface.
Alkali removal fluxes: Specifically formulated to react with dissolved sodium, calcium, and lithium (in Li-containing alloys) in the melt, forming compounds that float to the surface or transfer into a slag phase.
Covering Fluxes: Protecting Molten Aluminum from Oxidation
Covering flux is the most fundamental flux application in any aluminum melting operation. Its purpose is simple: prevent atmospheric oxygen and moisture from contacting the molten aluminum surface, thereby preventing continuous oxide skin formation and hydrogen pickup.
Composition of Covering Fluxes
Effective covering fluxes for aluminum must:
- Melt at or below the aluminum holding temperature (680–750°C for most alloys).
- Form a continuous, dense liquid layer that does not allow gas exchange between the melt and the atmosphere.
- Not react with aluminum to form undesirable compounds.
- Have low viscosity at operating temperature to flow and self-heal if disturbed.
- Be immiscible with liquid aluminum (so the flux layer stays on top).
The most common covering flux composition for standard aluminum alloys is a eutectic mixture of sodium chloride (NaCl) and potassium chloride (KCl) in approximately 50:50 or 40:60 molar ratio. The NaCl-KCl eutectic melts at approximately 660°C — just at or slightly below the aluminum melting point — providing a fully liquid protective blanket at normal holding temperatures.
This binary chloride system is effective, widely available, and relatively inexpensive. However, the plain NaCl-KCl system has limitations:
- It can introduce sodium into the melt through slow dissolution and reaction with the melt surface.
- It provides no inclusion removal function.
- It does not significantly reduce dross reactivity.
Adding small amounts of fluoride compounds (typically 5–15% cryolite, calcium fluoride, or magnesium fluoride) to the base NaCl-KCl covering flux improves performance in several ways: the fluoride components reduce the flux melting point further, lower flux viscosity at operating temperature (improving surface coverage), and provide some oxide film dissolving action at the flux-melt interface.
Covering Flux Application Method
Covering flux is applied by sprinkling or shoveling the flux powder or granules over the entire melt surface immediately after skimming, then allowing it to melt and spread. The application rate is typically 1–3 kg of flux per metric ton of aluminum held, with the actual rate adjusted to maintain a continuous, visible flux layer.
The flux layer should be inspected and refreshed after each metal addition, after sampling, and after any operation that disturbs the surface. A disrupted or thinned flux layer allows localized oxidation that generates inclusions even if the overall melt temperature and composition are correct.
Covering Flux Composition Examples
| Flux Designation | NaCl (%) | KCl (%) | CaF₂ (%) | Na₃AlF₆ (%) | MgCl₂ (%) | Primary Function |
|---|---|---|---|---|---|---|
| Standard covering | 50 | 50 | — | — | — | Basic surface protection |
| Fluoride-enhanced covering | 40 | 45 | 10 | 5 | — | Better fluidity, mild cleaning |
| Low-sodium covering | 20 | 60 | 10 | — | 10 | Reduced Na pickup |
| Magnesium alloy covering | 30 | 50 | 15 | — | 5 | Reduced Mg oxidation |
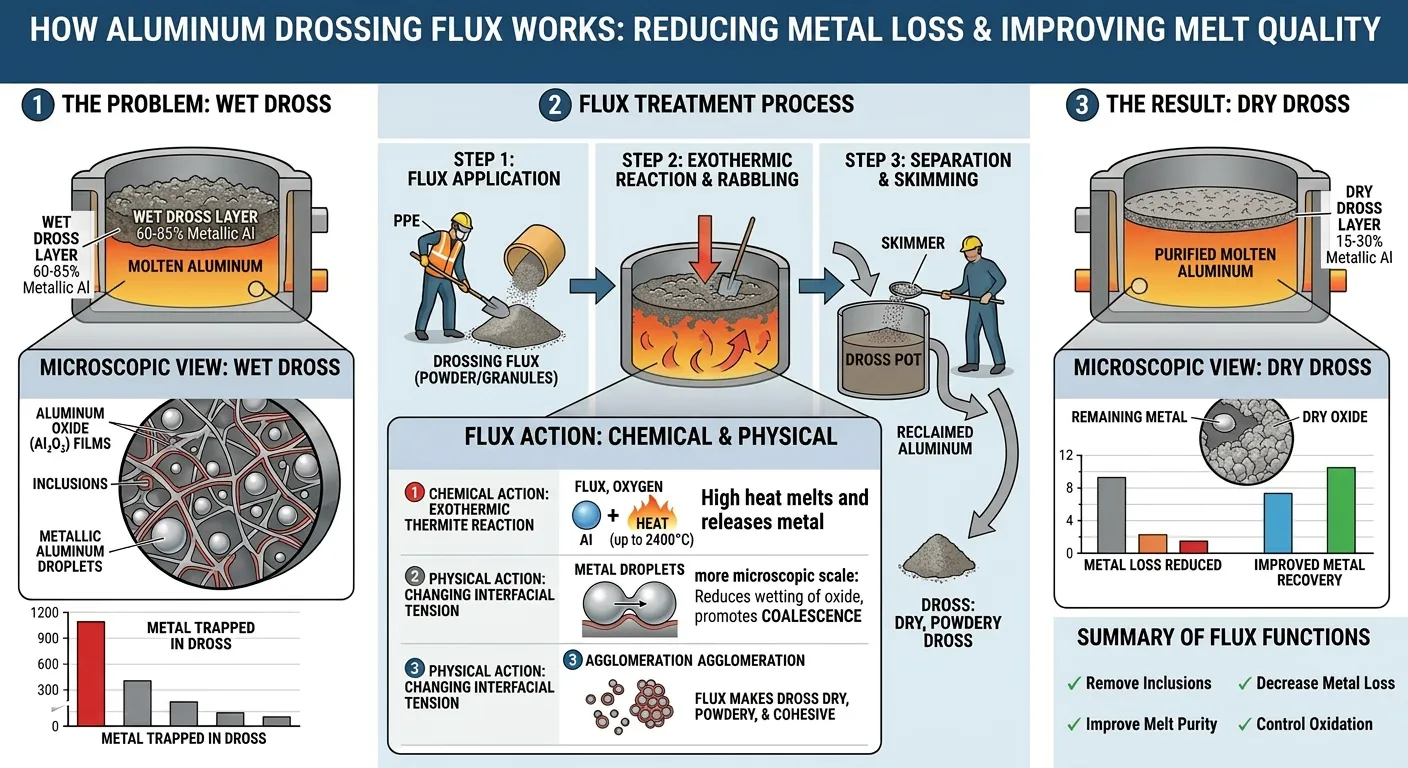
Drossing Fluxes: How They Separate Metal from Oxide
Dross is the accumulated layer of aluminum oxide, entrained metal, and other non-metallic compounds that forms on the surface of molten aluminum during holding, melting, and metal transfer. In primary aluminum production and secondary smelting operations, dross can represent 1–8% of the total metal volume — a significant material loss if not properly treated.
The Metal Recovery Problem in Dross
Dross from aluminum melting typically contains 30–70% metallic aluminum by weight, trapped in an oxide matrix. Without treatment, this metal is disposed of with the oxide fraction, representing direct metal loss and waste disposal cost. Drossing fluxes are specifically designed to liberate this trapped metal.
The mechanism works through surface tension modification. In untreated dross, the oxide particles are poorly wetted by liquid aluminum — the contact angle between liquid aluminum and the oxide surface is relatively high, meaning aluminum does not spread and flow through the oxide pore network efficiently. Drossing flux components (particularly fluoride compounds) adsorb at the aluminum-oxide interface and dramatically reduce the contact angle, allowing liquid aluminum to coalesce and flow out of the oxide matrix under gravity.
Drossing Flux Composition and Application
Drossing fluxes typically contain a higher proportion of reactive fluoride compounds than covering fluxes. Common compositions include:
| Component | Typical Range (%) | Function |
|---|---|---|
| KCl | 30–50 | Base flux, melting point adjustment |
| NaCl | 20–35 | Base flux, cost reduction |
| Na₃AlF₆ (cryolite) | 10–25 | Surface tension reduction, wetting |
| AlF₃ | 5–15 | Enhanced reactivity, fluoride activity |
| CaF₂ | 5–15 | Viscosity reduction, melting point |
| NaF | 0–10 | High reactivity, alkali removal |
Application procedure: after skimming the bulk dross to one side of the furnace, drossing flux is sprinkled over the dross layer (typically 5–15 kg per metric ton of dross), then the dross is raked and stirred to incorporate the flux throughout the mass. After 5–10 minutes of stirring and reaction time, the treated dross is noticeably drier and less metallic in appearance, and the metal content is reduced. Treated dross generates substantially less secondary dross (aluminum oxide skin) on the remaining metal surface than untreated dross during the skimming process.
Metal recovery improvement from drossing flux treatment ranges from 30–60% reduction in dross volume (meaning more metal stays in the furnace), with documented payback periods on flux cost typically measured in days rather than months in operations with significant dross generation.
Cleaning and Inclusion Removal Fluxes
Cleaning fluxes address the problem of suspended non-metallic inclusions within the melt bulk, as distinct from surface dross. These inclusions — primarily alumina films, spinel particles, titanium diboride agglomerates, and other non-metallic compounds — are dispersed throughout the melt volume and cannot be removed by surface skimming alone.
How Cleaning Fluxes Remove Inclusions
The mechanism of inclusion removal by flux involves two simultaneous processes:
Wetting and agglomeration: Flux components that have lower surface energy than aluminum oxide adsorb onto inclusion surfaces, lowering the interfacial tension between the inclusion and the flux phase. This promotes agglomeration of small inclusions into larger clusters, which then have sufficient buoyancy to float to the surface.
Chemical reaction: Some flux components (particularly fluoride compounds) react directly with aluminum oxide, forming aluminum fluoride and releasing oxide lattice oxygen. This dissolution reaction reduces the size of oxide inclusions directly and promotes their transfer from the melt to the salt flux phase.
Research published by Groteke and Neff in AFS Transactions (1993) demonstrated that flux treatment with fluoride-containing salts reduced inclusion content measured by PoDFA by 40–65% in A356 alloy, with the improvement strongly correlated with the fluoride activity of the flux.
Reactive vs. Non-Reactive Cleaning Fluxes
Reactive cleaning fluxes contain higher proportions of fluoride compounds and generate visible gas evolution (from fluoride-oxide reactions) when stirred into the melt. They provide stronger inclusion removal but must be used carefully because:
- Excessive fluoride activity can attack furnace refractory linings.
- Gas evolution creates turbulence that generates new oxide films if melt agitation is not controlled.
- Some reactive fluxes introduce sodium or other impurities.
Non-reactive (physical) cleaning fluxes rely primarily on density separation and wetting modification without significant chemical reaction with the melt. They are gentler on refractory and generate no gas, but provide less aggressive inclusion removal.
Inclusion-Type Compatibility
| Inclusion Type | Physical Cleaning Flux Effectiveness | Reactive (Fluoride) Flux Effectiveness |
|---|---|---|
| Alumina films (Al₂O₃) | Moderate | Good to Excellent |
| Spinel (MgAl₂O₄) | Fair | Good |
| TiB₂ agglomerates | Fair | Moderate |
| NaCl/KCl salt particles | Good (dissolves in flux) | Excellent |
| Refractory particles | Poor | Poor |
| Carbides (Al₄C₃) | Poor | Moderate |
Improving the efficiency of aluminum refining agent deployment can be achieved through the use of AdTech’s Flux Injection Machine.
Degassing Fluxes and Their Hydrogen Removal Function
Historically, reactive compounds — particularly hexachloroethane (C₂Cl₆) in tablet form — were the primary method for hydrogen removal from aluminum melts. These “degassing fluxes” generated chlorine gas when they dissolved in the melt:
C₂Cl₆ → 2C + 3Cl₂
The chlorine bubbles rising through the melt collected dissolved hydrogen (forming HCl) and carried it to the surface, reducing melt hydrogen content by 30–50%.
Current Status of Degassing Flux Use
Hexachloroethane and similar reactive halogen-generating compounds face severe regulatory pressure globally:
Environmental concerns: Chlorine and HCl gas generation during treatment requires fume extraction systems. Perchloroethylene and other organochlorine byproducts of hexachloroethane reaction are classified as hazardous air pollutants under the US Clean Air Act.
Effectiveness limitations: At 30–50% hydrogen reduction, reactive degassing flux significantly underperforms rotary inert gas degassing (50–80% reduction) and is not adequate for quality-sensitive applications.
European restrictions: Several EU member states have restricted or prohibited hexachloroethane use in aluminum processing. Alternative degassing methods are strongly preferred in European operations.
Current use: Hexachloroethane tablets remain in use in small foundries in developing markets where rotary degassing equipment is not justified economically, and for emergency degassing when primary equipment is unavailable. For any operation with quality specifications requiring hydrogen below 0.12 ml/100g Al, rotary inert gas degassing with argon or nitrogen is the standard approach — not reactive degassing flux.
At AdTech, we supply chlorine-containing gas mixtures (typically 2–5% Cl₂ in argon) for use through rotary degassing equipment rather than solid degassing flux tablets. The Cl₂ in the gas mixture provides the inclusion agglomeration benefit of chlorine chemistry without the environmental problems of solid hexachloroethane.

Alkali Removal Fluxes: Sodium and Calcium Treatment
Sodium (Na) and calcium (Ca) impurities in aluminum melts cause specific problems that standard covering or cleaning fluxes do not adequately address.
Why Alkali Metals Are Harmful in Aluminum
Sodium effects: At concentrations above approximately 5–10 ppm, sodium modifies the silicon phase morphology in Al-Si alloys — an effect that is intentional in sodium-modified alloys but harmful in others. In high-Mg alloys, sodium promotes hot tearing and grain boundary embrittlement. In wire rod for electrical applications, sodium above 5 ppm reduces conductivity and wire drawing performance.
Calcium effects: Calcium above approximately 3–5 ppm can modify grain structure in ways that reduce mechanical properties and cause surface quality problems in wrought products. Calcium is also associated with increased porosity susceptibility in some alloy systems.
Sources of sodium contamination include: salt flux residues from previous furnace operations, some grades of recycled scrap (particularly contaminated with glass or ceramic), master alloy additions where sodium is a minor impurity, and poorly maintained degassing flux tablet storage (hexachloroethane tablets can introduce sodium from moisture-degraded tablet binders).
Alkali Removal Flux Mechanism
Alkali removal fluxes typically contain fluoride compounds with high thermodynamic affinity for sodium and calcium. The most effective alkali removal agents are:
AlF₃ (aluminum fluoride): Reacts with dissolved sodium:
3Na + AlF₃ → Al + 3NaF (transferred to salt flux phase)
The reaction is thermodynamically favorable at aluminum melt temperatures and proceeds rapidly when the flux is in contact with the melt.
Chlorine gas (through rotary degassing): Chlorine reacts with dissolved sodium and calcium to form NaCl and CaCl₂, which transfer to the salt phase floating on the melt surface. This is one of the primary reasons that small chlorine additions to rotary degassing gas (2–5% Cl₂) are specified for operations producing high-specification aluminum — the chlorine addresses alkali removal simultaneously with hydrogen reduction.
Alkali Metal Removal Efficiency
| Treatment Method | Na Removal (% of initial) | Ca Removal (% of initial) | Treatment Time |
|---|---|---|---|
| AlF₃-containing flux, stirring | 60–80% | 50–70% | 5–15 minutes |
| Rotary degassing, Ar only | 20–35% | 15–30% | 15–30 minutes |
| Rotary degassing, Ar + 3% Cl₂ | 75–90% | 65–85% | 15–30 minutes |
| Reactive flux + rotary degassing | 85–95% | 75–90% | Combined treatment |
For EC-grade aluminum (1350 alloy for electrical conductor applications) where sodium must be below 5 ppm, the combined treatment of alkali-reactive flux followed by Ar + Cl₂ rotary degassing is the standard approach in premium operations.
Flux Composition Tables and Chemical Systems
Comprehensive Flux Types and Compositions Reference
| Flux Type | Base System | Key Additives | Application Rate | Operating Temp Range |
|---|---|---|---|---|
| Standard covering | 50% NaCl, 50% KCl | None | 1–3 kg/t | 660–800°C |
| Fluoride covering | 40% NaCl, 45% KCl | 10–15% CaF₂ | 1–3 kg/t | 660–800°C |
| Drossing flux | 35% KCl, 25% NaCl | 25% Na₃AlF₆, 15% AlF₃ | 5–15 kg/t dross | 680–760°C |
| Cleaning flux (moderate) | 45% KCl, 35% NaCl | 15% CaF₂, 5% NaF | 2–5 kg/t | 700–760°C |
| Cleaning flux (reactive) | 35% KCl, 25% NaCl | 20% Na₃AlF₆, 15% AlF₃, 5% NaF | 3–8 kg/t | 700–760°C |
| Alkali removal flux | 30% KCl, 20% NaCl | 30% AlF₃, 20% CaF₂ | 3–10 kg/t | 700–760°C |
| Degassing flux (legacy) | C₂Cl₆ tablets | — | 0.5–2 kg/t | 680–750°C |
| Magnesium alloy flux | 35% KCl, 35% MgCl₂ | 20% NaCl, 10% CaF₂ | 2–5 kg/t | 680–760°C |
Melting Points of Common Flux Components
| Compound | Chemical Formula | Melting Point (°C) | Function in Flux |
|---|---|---|---|
| Sodium chloride | NaCl | 801°C | Base flux component |
| Potassium chloride | KCl | 770°C | Base flux component (lowers eutectic) |
| NaCl-KCl eutectic | — | ~660°C | Lowest melting point of binary system |
| Calcium fluoride | CaF₂ | 1418°C | Viscosity reduction, wetting |
| Cryolite | Na₃AlF₆ | 1009°C | Reactive, surface tension reduction |
| Aluminum fluoride | AlF₃ | 1291°C (subl.) | Alkali removal, reactive |
| Sodium fluoride | NaF | 993°C | High reactivity, fluoride activity |
| Magnesium chloride | MgCl₂ | 714°C | Mg alloy flux component |
| Magnesium fluoride | MgF₂ | 1263°C | Covering flux modifier |
Flux Selection by Alloy Series
| Alloy Series | Primary Concern | Recommended Flux Type | Key Restriction |
|---|---|---|---|
| 1xxx (pure Al, EC grade) | Na, Ca impurity, inclusions | Alkali removal + covering | Minimize Na addition from flux |
| 2xxx (Al-Cu) | Inclusions, oxide films | Cleaning flux + covering | No high-Na flux |
| 3xxx (Al-Mn) | Inclusions, Fe-Si particles | Covering + moderate cleaning | Standard |
| 5xxx (Al-Mg, <3% Mg) | MgO inclusions, surface oxide | Fluoride covering | Reduced Na |
| 5xxx (Al-Mg, >3% Mg) | Rapid oxidation, MgO | Magnesium-specific covering | Avoid high-NaCl flux |
| 6xxx (Al-Mg-Si) | Inclusions, TiB₂ agglomerates | Cleaning + covering | Standard |
| 7xxx (Al-Zn-Mg) | Inclusions, alkali removal | Reactive cleaning + alkali removal | No excess fluoride |
| A356/A380 (casting) | Hydrogen, inclusions | Covering + drossing | Minimize new inclusion introduction |
How to Choose the Right Aluminum Flux for Your Application
Selecting the correct flux requires matching the flux’s primary function to the dominant melt quality problem in the specific operation. Using the wrong flux type wastes material, may introduce new problems, and gives a false sense of security about melt quality.

Step 1: Identify Your Primary Melt Quality Problem
Conduct RPT (Reduced Pressure Test) to assess combined hydrogen and bifilm content. Use PoDFA sampling to quantify inclusion type and quantity. Measure sodium content by emission spectrometry if alkali contamination is suspected. Only with this diagnostic information can flux selection be genuinely optimized rather than based on generic recommendations.
Step 2: Match Flux Function to Diagnosed Problem
| Diagnosed Problem | Primary Flux Solution | Secondary Treatment |
|---|---|---|
| High dross volume with high metal content | Drossing flux | Improve covering flux coverage |
| Suspended inclusions in bulk melt | Cleaning flux (fluoride-containing) | Downstream ceramic foam filtration |
| Rapid surface oxidation, high dross generation rate | Better covering flux, applied more frequently | Reduce melt surface turbulence |
| High sodium or calcium content | Alkali removal flux, Cl₂ addition to rotary degassing | Investigate source of alkali contamination |
| High hydrogen, porosity in castings | Rotary degassing (primary), Cl₂ gas addition | Covering flux to reduce re-absorption |
| General inclusion burden from scrap | Combined cleaning + covering approach | Two-stage CFF filtration |
Step 3: Consider Alloy-Specific Restrictions
High-magnesium alloys (5xxx with Mg >3%): Standard NaCl-KCl covering flux reacts with magnesium in the melt, introducing sodium and potentially disrupting the magnesium balance. Magnesium-specific flux formulations using MgCl₂-KCl-NaCl systems with minimal sodium activity are required.
Electrical conductor grade (1350 alloy): Any flux containing significant sodium must be used with caution. Post-flux treatment sodium levels must be verified analytically. The alkali removal function should be performed before the covering flux is applied, and the covering flux should be a low-sodium formulation.
Low-silicon alloys: Some fluoride flux components promote silicon reduction from refractory and from the flux system itself. In very low-silicon applications, verify that the flux does not introduce silicon.
Step 4: Optimize Application Procedure
The most common flux application failure is using the correct flux product but applying it incorrectly:
- Apply too little flux and coverage is incomplete, allowing continued oxidation.
- Apply too much flux and the thick flux layer becomes difficult to skim cleanly, introducing salt inclusions.
- Apply flux without adequate stirring in cleaning applications, leaving most of the melt volume untreated.
- Allow flux to become contaminated with water (salt flux absorbs moisture rapidly from humid air, which can cause violent spattering when introduced to the melt).
Safety, Environmental Regulations, and Flux Waste Management
Occupational Hazards of Aluminum Flux Use
Aluminum fluxes present several occupational hazards that require active management:
Hydrogen chloride (HCl) fume generation: When chloride fluxes contact moisture (in the air, on tools, or from wet charge material), they generate HCl fumes. OSHA PEL for HCl is 5 ppm ceiling. Operations using chloride fluxes require local exhaust ventilation.
Fluoride fume generation: Fluoride-containing fluxes generate hydrogen fluoride (HF) fumes, particularly when heated. HF is acutely toxic at low concentrations (OSHA PEL 3 ppm TWA, 6 ppm STEL). Fluoride flux use requires respiratory protection and fume extraction.
Molten salt spatter: Introducing flux to the melt surface requires controlled application to prevent steam explosion from moisture in the flux or on the application tool. All flux must be stored dry and preheated before use in any application involving immersion below the melt surface.
Thermal burns: Working with molten aluminum and molten salt flux at 700–760°C presents severe burn risk. All personnel must wear appropriate PPE including face shield, heat-resistant gloves, and heat-resistant clothing.
Environmental Regulations Affecting Flux Choice
| Regulation | Region | Impact on Flux Selection |
|---|---|---|
| EU Regulation 1907/2006 (REACH) | European Union | Restrictions on certain fluoride compounds; cryolite registration required |
| Clean Air Act (NESHAP) | USA | Limits on HCl and HF emissions from secondary aluminum operations |
| EU F-Gas Regulation | European Union | Limits on halogenated compounds in industrial processes |
| China GB Standards | China | Maximum emission limits for HF and Cl₂ from aluminum processing |
| RoHS Directive | EU | Affects flux composition in aluminum for electronic applications |
Flux Waste (Salt Slag) Disposal
Used aluminum flux and associated dross (called salt slag or black dross) contains chloride and fluoride salts mixed with aluminum oxide and residual metal. This material is classified as hazardous waste in most regulatory jurisdictions due to its:
- Leachability of chlorides and fluorides into groundwater.
- Potential generation of ammonia (from nitride impurities) when wet.
- Heavy metal content from aluminum alloy impurities.
Salt slag must be disposed of at licensed hazardous waste facilities or processed through salt slag recycling operations that recover the salt fraction for reuse and the aluminum oxide fraction for other applications. Several commercial salt slag processing facilities operate in Europe, North America, and East Asia. The environmental liability from improper salt slag disposal significantly exceeds the material cost of the original flux — this is one reason that minimizing flux use through optimized application is both economically and environmentally desirable.
Frequently Asked Questions About Flux Used for Aluminum
1: What is the most common flux used for aluminum melting?
The most widely used aluminum flux is a mixture of sodium chloride (NaCl) and potassium chloride (KCl) in approximately equal proportions by weight, often with small additions of calcium fluoride (CaF₂) or cryolite (Na₃AlF₆). This NaCl-KCl base system forms a eutectic that melts at approximately 660°C — just at or below aluminum’s melting point — creating a liquid protective blanket on the melt surface that prevents oxidation and hydrogen pickup. This covering flux is used in virtually every aluminum foundry and smelting operation worldwide as the baseline melt protection. More specialized fluxes (drossing agents, cleaning fluxes, alkali removal fluxes) are added for specific treatment objectives beyond basic surface protection. The NaCl-KCl system’s dominance reflects its low cost, wide availability, and proven performance across the full range of commercial aluminum alloys.
2: Can I use borax or other common fluxes for aluminum?
No — borax (sodium tetraborate, Na₂B₄O₇) is a flux used for soldering and brazing of metals including aluminum at lower temperatures, but it is not appropriate as a melt treatment flux for molten aluminum foundry work. Borax has a melting point of approximately 743°C, which is within the range of aluminum melting temperatures, but it reacts with aluminum to form aluminum boride compounds and introduces boron contamination into the melt. In grain refiner chemistry, boron is a controlled addition — uncontrolled boron from borax flux use would disrupt the carefully managed grain refinement of commercial aluminum alloys. The correct fluxes for molten aluminum production are chloride-fluoride salt systems as described in this article. For aluminum brazing and soldering (a different process from melt treatment), noncorrosive flux systems based on fluoride compounds are used, such as Nocolok (potassium fluoroaluminate) for furnace brazing of aluminum heat exchangers.
3: What is the difference between covering flux and cleaning flux for aluminum?
Covering flux and cleaning flux serve different functions and are applied differently. Covering flux is spread over the entire melt surface to prevent contact between liquid aluminum and atmospheric oxygen — it physically isolates the melt surface. It is typically applied at a low rate (1–3 kg per metric ton of aluminum) and maintained throughout the entire holding period. Cleaning flux is designed to react with and remove suspended non-metallic inclusions from within the melt bulk — it must be stirred or injected into the melt to make contact with inclusions throughout the metal volume. Cleaning fluxes contain higher proportions of reactive fluoride compounds and are applied at higher rates (2–8 kg per metric ton) but less frequently — typically once per furnace charge rather than continuously. Some products marketed as “combined” flux attempt to perform both functions simultaneously, but in our experience at AdTech, the best results come from using a dedicated covering flux for continuous surface protection and a separate cleaning flux for periodic bulk treatment.
4: How does flux remove inclusions from molten aluminum?
Flux removes inclusions from molten aluminum through two complementary mechanisms: surface tension modification and chemical reaction. The surface tension mechanism works because fluoride compounds in the flux adsorb at the interface between aluminum oxide inclusion particles and the surrounding metal, lowering the interfacial energy and promoting inclusion agglomeration into larger clusters. Larger clusters have greater buoyancy relative to their drag and rise to the melt surface more readily than small individual inclusions. The chemical reaction mechanism involves direct dissolution of aluminum oxide by fluoride species — particularly AlF₃ and cryolite — which react with Al₂O₃ to form aluminum oxyfluoride and related compounds that partition preferentially into the salt flux phase rather than remaining in the metal. Both mechanisms require the flux to be in intimate contact with the inclusions, which is why cleaning flux must be stirred thoroughly into the melt rather than simply floated on the surface.
5: Is flux required if I use rotary degassing and ceramic foam filtration?
Flux is not entirely replaced by rotary degassing and ceramic foam filtration, but its role changes significantly when these systems are in place. Rotary degassing handles the dissolved hydrogen function that reactive degassing flux (hexachloroethane) previously addressed, and ceramic foam filtration removes inclusions that cleaning flux would otherwise have to treat. However, covering flux remains necessary regardless of degassing and filtration systems — the melt surface continuously generates new oxide as long as it is exposed to atmosphere, and this surface oxide must be physically managed with a covering flux blanket to prevent it from entering the melt as inclusions. Drossing flux for metal recovery from accumulated dross is also still needed. What changes when rotary degassing and CFF are available is that the reactive cleaning function of fluxes (particularly reactive fluoride fluxes) becomes less critical, reducing overall flux consumption and the associated waste management challenges.
6: What flux is used for aluminum with high magnesium content (5xxx series)?
High-magnesium aluminum alloys (5xxx series with Mg above 3%, such as 5083 and 5182) require specially formulated flux that avoids introducing significant amounts of sodium, which reacts with magnesium and causes problems. Standard NaCl-KCl covering flux contains substantial sodium that can exchange with magnesium in the melt surface oxide, introducing sodium impurity and potentially disrupting the magnesium balance. The appropriate covering flux for high-Mg alloys uses magnesium chloride (MgCl₂) as a primary component alongside KCl, with minimal NaCl content — for example, a 35% MgCl₂, 50% KCl, 15% NaCl mixture. This low-sodium formulation provides adequate surface protection without the sodium exchange reaction. Additionally, for high-Mg alloys, the flux layer must be maintained more carefully than for low-Mg alloys because the magnesium in the melt oxidizes rapidly and generates MgO inclusions that are more difficult to remove than Al₂O₃. More frequent flux refreshing and gentle melt handling are required.
7: Can flux be used to remove hydrogen from aluminum or is degassing equipment needed?
Reactive degassing flux (hexachloroethane tablets) can remove hydrogen from aluminum, but only at limited efficiency (30–50% reduction) and with significant environmental and safety drawbacks. For most quality-sensitive applications, rotary inert gas degassing with argon or nitrogen is the required method for hydrogen removal because it achieves 50–80% hydrogen reduction consistently, can be precisely controlled, and does not generate toxic chlorine compounds that require fume extraction. The addition of small amounts of chlorine gas (2–5% Cl₂) to the rotary degassing argon provides additional benefits including inclusion agglomeration and alkali metal removal — this approach combines the best of both flux chemistry and mechanical degassing without the problems of solid flux tablets. At AdTech, we recommend reactive solid degassing flux only as an emergency backup measure when primary degassing equipment is unavailable, or in very small operations where rotary equipment cannot be economically justified.
8: What flux is used for aluminum soldering and brazing versus foundry fluxes?
These are completely different flux systems serving different processes. For furnace brazing of aluminum components (such as automotive heat exchangers), the standard flux system is potassium fluoroaluminate (K₁₋₃AlF₄₋₆), commercially known as Nocolok or equivalent products. This flux becomes active above approximately 560°C, disrupts the oxide layer on aluminum surfaces during brazing, and allows the brazing filler metal (typically Al-Si eutectic alloy) to wet and flow into the joint. For torch brazing, aluminum brazing fluxes based on similar fluoride chemistries are applied as paste or powder to the joint area before heating. For soft soldering of aluminum (at lower temperatures using zinc or tin-based solders), aggressive fluxes based on organic acids or zinc chloride are used. None of these brazing or soldering fluxes are appropriate for foundry melt treatment — they are designed for surface wetting of solid aluminum, not for bulk treatment of large volumes of liquid metal at 700–760°C.
9: How much flux should I add to molten aluminum, and how often?
Application rates depend on flux type and operating conditions. Covering flux: 1–3 kg per metric ton of aluminum held, applied after each skimming operation and after any event that disturbs the melt surface (metal additions, sampling, tool immersion). In humid conditions or when the melt is held for extended periods, covering flux should be refreshed every 30–60 minutes. Cleaning flux: 2–8 kg per metric ton of aluminum, applied once per furnace charge during the treatment phase, with thorough stirring for 5–10 minutes to distribute the flux through the melt volume. Drossing flux: 5–15 kg per metric ton of dross, applied directly to the dross layer and worked in with a rake. Alkali removal flux: 3–10 kg per metric ton, with treatment time of 10–20 minutes of stirring before skimming. Over-application of any flux type increases salt inclusion risk (flux particles becoming entrapped in the metal) and increases waste disposal volume and cost. Systematic optimization of flux consumption — through regular RPT testing to verify effectiveness and furnace records to track consumption versus quality outcomes — consistently reduces both flux cost and waste generation.
10: What certifications or quality standards should flux for aluminum meet?
Industrial aluminum flux for foundry and casthouse applications should meet or be verified against the following quality and safety standards. ISO 9001 certification of the manufacturer ensures consistent production quality and traceability. Chemical composition should be verified by the manufacturer’s analytical laboratory per each production batch, with certificates of conformance available for every shipment. Flux purity is particularly important for moisture content — water in flux above approximately 0.2% can cause violent spattering when the flux is applied to the melt. Heavy metal content (lead, cadmium, mercury) must be confirmed below specified limits, particularly for flux used in aluminum destined for food contact or automotive structural applications. Safety Data Sheet (SDS) per GHS requirements must be current and include emergency procedures for skin and eye contact with molten salt and for inhalation of fluoride fumes. REACH compliance documentation is required for European market procurement. For flux used in aerospace aluminum production, the manufacturer should provide full traceability records and the flux composition must be verified compatible with the specific alloy’s impurity specifications — particularly for sodium and calcium content that the flux itself might introduce.
Summary: Choosing the Right Flux for Your Aluminum Operation
The flux used for aluminum is not a single product but a family of specialized chemical treatments, each targeting a specific melt quality problem. The most important principle in flux selection is matching the flux function to the diagnosed problem rather than applying a generic “aluminum flux” without identifying what the actual quality issue is.
Covering flux (NaCl-KCl base with fluoride modifiers) is the universal baseline — no aluminum holding operation should proceed without adequate surface coverage. Drossing flux improves metal recovery from dross and reduces waste volume in operations with significant dross generation. Cleaning flux reduces suspended inclusion content in the bulk melt, working synergistically with downstream ceramic foam filtration. Alkali removal flux addresses sodium and calcium contamination that standard fluxes cannot resolve. And rotary degassing with controlled gas chemistry (argon or nitrogen with optional Cl₂ addition) handles the hydrogen removal function that reactive solid flux once attempted to serve.
The optimal melt treatment system combines these elements in the right sequence — covering flux for continuous protection, periodic cleaning flux for bulk inclusion management, alkali removal treatment when composition requires it, and rotary degassing as the primary tool for hydrogen control. Each component has a specific role, and the system performs best when all roles are filled correctly.
At AdTech, we supply the full range of aluminum melt treatment products, including flux formulations, ceramic foam filters, and rotary degassing equipment, and our application engineering team supports customers in designing melt treatment systems that achieve their quality targets at the lowest total treatment cost.
This article was prepared by the AdTech technical editorial team based on primary foundry experience, published metallurgical research, and direct application monitoring at aluminum casting facilities. Key references include work by Groteke and Neff (AFS Transactions, 1993) and industry standard practices documented by the Aluminum Association and European Aluminium. Content is reviewed annually.
Last updated: 2026 | AdTech Technical Resource Library.

