hydrogen dissolves far more readily in molten aluminum than in solid aluminum, typically by roughly 10 to 20 times near the melting range, and that sudden drop in solubility during freezing is the direct reason untreated aluminum develops pinholes, gas porosity, blisters, and internal voids. This single metallurgical fact explains why melt quality control is so important in aluminum casting. If a plant wants cleaner billet, slab, wheel, die casting, or gravity casting output, it must limit moisture pickup, reduce turbulence, verify dissolved hydrogen, and use effective degassing before the metal enters the mold. In short, the contrast between liquid-state hydrogen capacity and solid-state hydrogen capacity is not a textbook detail. It is one of the main levers behind scrap rate, pressure tightness, surface finish, and downstream mechanical performance.
If your project requires the use of Degassing Molten Aluminum Unit, you can contact us for a free quote.
Hydrogen is the only gas with meaningful solubility in aluminum under normal foundry and casthouse conditions. That statement appears in most technical discussions on aluminum melt treatment, yet the practical meaning is often underexplained. Engineers know hydrogen matters. Operators know porosity appears when melt treatment slips. Buyers know rotary degassing units, graphite rotors, analyzers, and filtration systems cost money. What ties all of these decisions together is the thermodynamic gap between hydrogen in liquid aluminum and hydrogen in solid aluminum. Once this relationship is clear, decisions on degassing, testing, alloy handling, and equipment selection become easier and more rational.
Hydrogen Solubility in Pure Aluminum,Degassing Method
Why is hydrogen much more soluble in molten aluminum than in solid metal?
The short answer is structural freedom. Liquid aluminum has a disordered atomic arrangement, while solid aluminum has an ordered crystalline lattice. Hydrogen atoms can exist more easily in the liquid state because the melt offers more temporary interstitial space and greater atomic mobility. Once aluminum solidifies, the crystal lattice becomes much less accommodating to hydrogen.
What happens at the atomic level?
In the melt, hydrogen exists in atomic form after dissociation at the metal surface. It diffuses into the liquid and remains dissolved according to thermodynamic equilibrium. In solid aluminum, the available lattice sites are limited and energetically less favorable. That means the metal can no longer hold the same amount of hydrogen.
This is the root of the problem during casting:
- Liquid aluminum can hold relatively high dissolved hydrogen.
- Solid aluminum can hold very little.
- The excess must go somewhere during freezing.
- If it cannot escape, it forms pores.
Why does this matter so much during solidification?
Solidification is not instantaneous. A casting or billet freezes over time, forming dendrites and pockets of remaining liquid. Hydrogen rejected from the advancing solid accumulates in the last liquid regions. The local gas concentration rises rapidly. If pressure and nucleation conditions allow, pores form in interdendritic areas.
That means the danger is not only “high hydrogen in the melt.” The danger is hydrogen plus solidification.
Is this behavior unique to aluminum?
Hydrogen solubility differences also matter in other metals, yet aluminum is especially sensitive because:
- Hydrogen is the dominant dissolved gas concern.
- Oxide films and bifilms provide easy pore nucleation sites.
- Many aluminum products need tight surface and internal quality.
- Recycled charge and moisture exposure can raise gas pickup quickly.
How large is the hydrogen solubility difference between molten aluminum and solid aluminum?
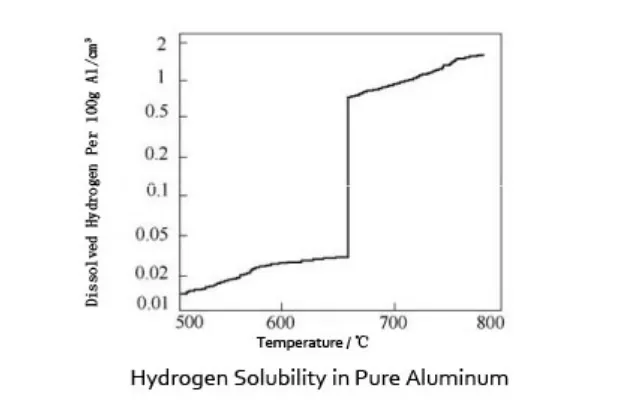
Near the melting point, the difference is dramatic. Depending on data source, alloy family, and unit system, liquid aluminum near casting temperature can dissolve roughly 10 to 20 times more hydrogen than solid aluminum at the freezing boundary.
Approximate hydrogen solubility comparison
| State of aluminum | Typical temperature range | Approximate hydrogen solubility at equilibrium | Practical meaning |
|---|---|---|---|
| Molten aluminum | Around 700°C to 750°C | About 0.6 to 0.8 cm³ H2 per 100 g Al | Melt can retain significant dissolved hydrogen |
| Molten aluminum near liquidus | Around 660°C to 700°C | About 0.5 to 0.7 cm³ H2 per 100 g Al | Still much higher than the solid state |
| Solid aluminum near freezing | Around 660°C | About 0.03 to 0.04 cm³ H2 per 100 g Al | Very low hydrogen capacity |
| Solid aluminum at room temperature | Around 20°C to 25°C | Extremely low, close to negligible in practical terms | Hydrogen cannot remain dissolved at prior liquid levels |
These values are representative ranges used in industrial discussion. Exact numbers vary with reference source, alloy chemistry, and test method. The critical point is not the last decimal. The critical point is the steep gap across the liquid-to-solid transition.
Why engineers often quote a ratio instead of a single value
Because the numbers shift with temperature, pressure, and alloying additions, many metallurgists describe the issue by ratio. Near the melting point, the liquid state may hold around 15 to 20 times more hydrogen than the solid state. That ratio explains why even a melt that appears manageable in the furnace can create serious porosity when cast.
Useful unit conversion for plant discussions
Different plants report hydrogen in different units. That creates confusion during audits, process transfer, or supplier discussion.
| Unit expression | Meaning | Approximate relation |
|---|---|---|
| cm³ H2 / 100 g Al | Common technical solubility unit | Widely used in metallurgy references |
| mL H2 / 100 g Al | Same practical meaning as cm³ / 100 g | 1 mL = 1 cm³ |
| ppm by weight | Mass-based hydrogen content | Roughly, 1 mL H2 / 100 g Al is about 0.89 ppm H |
| Density index | Relative gas tendency indicator | Indirect, not a direct solubility unit |
Many misunderstandings in plants come from mixing these units without conversion.
What thermodynamic rule describes hydrogen dissolution in molten aluminum?
The most common starting point is Sieverts’ law. In simple terms, dissolved hydrogen concentration in the melt is proportional to the square root of the hydrogen partial pressure above the metal, multiplied by a temperature-dependent constant.
Simplified expression
[H]=K(T)×√PH2
Where:
- [H] is dissolved hydrogen concentration.
- K(T) is a temperature-dependent solubility constant.
- √PH2 is the partial pressure of hydrogen gas.
What this means in plant language
If the atmosphere above the melt contains more hydrogen potential, often due to moisture, hydrocarbons, or combustion products, hydrogen pickup becomes easier. If melt temperature rises, hydrogen solubility in the liquid also tends to rise. That is why hot, wet, turbulent metal is a perfect recipe for gas problems.
Why moisture matters even more than many plants expect
Foundries often say “water is the enemy of molten aluminum,” and this is not a slogan. It is a thermodynamic reality. Water vapor near hot metal dissociates, enabling hydrogen absorption. Sources include:
- Damp charge material.
- Wet tools
- Unheated ladles.
- Condensation on launder covers.
- Humid combustion gases.
- Leaks near cooling circuits.
A plant may degas carefully, yet still struggle with gas porosity if moisture control is poor.
What happens to dissolved hydrogen when aluminum changes from liquid to solid?
When solidification starts, hydrogen becomes less soluble in the newly formed solid. The advancing solid rejects hydrogen into the remaining liquid. This raises local hydrogen concentration in interdendritic channels and the last-freezing zones.
Why pore nucleation occurs in the mushy zone
During freezing, several conditions develop at the same time:
- Remaining liquid volume shrinks.
- Hydrogen concentration rises in that remaining liquid.
- Feeding becomes more difficult.
- Oxide films and inclusions may act as nucleation sites.
- Pressure conditions inside the mushy zone become favorable for pore growth.
If the gas cannot diffuse out, it nucleates pores. These pores may be spherical, elongated, interdendritic, or linked with shrinkage cavities.
Gas porosity and shrinkage porosity often interact
Plants sometimes separate gas porosity and shrinkage porosity as if they are unrelated. In reality, they often interact. Hydrogen can enlarge voids that would otherwise remain small. Poor feeding can amplify gas-related defects. The resulting porosity network may reflect both mechanisms.
Why bifilms make the problem worse
Oxide films folded into the melt, often called bifilms in aluminum metallurgy, can trap air or create crack-like interfaces. These internal surfaces are ideal sites for hydrogen pore nucleation. This is why turbulence control matters almost as much as degassing.
Solidification sequence and hydrogen behavior
| Stage | Hydrogen condition | Defect risk |
|---|---|---|
| Fully liquid melt | Hydrogen remains dissolved if under solubility limit | Hidden gas load, not always visible |
| Early freezing | Solid rejects hydrogen into remaining liquid | Local hydrogen enrichment begins |
| Mushy zone | Interdendritic liquid becomes hydrogen-rich | Pore nucleation and growth |
| Final solidification | Escape paths close | Porosity becomes trapped inside the metal |
| Cooling to room temperature | Solid hydrogen solubility becomes extremely low | Existing pores remain, cannot vanish by themselves |
Where does hydrogen come from in real aluminum melting and holding operations?
Most production hydrogen problems come from moisture, not from mysterious chemistry. Hydrogen pickup is usually traceable to handling discipline, furnace atmosphere, or raw material condition.
Common hydrogen sources in aluminum plants
| Source | How hydrogen enters the melt | Typical plant symptom |
|---|---|---|
| Wet scrap or returns | Moisture dissociates at hot metal contact | Rapid hydrogen rise after charging |
| Damp tools and skimmers | Water contact with melt surface | Local flare, dross, gas pickup |
| Unpreheated ladles and launders | Condensation or residual moisture | Variable gas level between heats |
| Humid furnace atmosphere | Water vapor interacts with exposed melt | Slow but persistent pickup |
| Fluxes with moisture contamination | Hydrogen source during treatment | Inconsistent degassing performance |
| Burner products | Combustion creates water vapor | High gas tendency in poorly ventilated furnaces |
| Turbulent transfer | Constant surface renewal exposes fresh metal | More oxidation and gas absorption |
Why recycled charge often raises the risk
Recycled aluminum can be excellent feedstock, yet it often carries oil, paint, oxide, and variable moisture exposure history. If preparation and drying are inconsistent, hydrogen pickup risk rises significantly.
Why holding time matters
Long holding at elevated temperature gives hydrogen more time to enter the melt, especially when the surface is exposed and the atmosphere is humid. This is one reason good plants do not rely on late-stage emergency degassing alone. They also reduce pickup upstream.
Which process conditions increase hydrogen solubility or hydrogen pickup the most?
Hydrogen behavior depends on more than one variable. Temperature matters, but so do atmosphere, exposure time, turbulence, and alloy chemistry.
Key process variables and their effect
| Variable | Effect on dissolved hydrogen | Production consequence |
|---|---|---|
| Higher melt temperature | Tends to increase hydrogen solubility | More hydrogen can remain in the liquid before casting |
| High humidity or water vapor | Raises hydrogen pickup potential | Gas level rises quickly |
| Long holding time | More time for absorption | Greater porosity risk if untreated |
| Turbulent pouring or transfer | Constantly renews reactive surface | More hydrogen pickup and more oxide entrainment |
| Dirty furnace surface and dross disturbance | Exposes metal to oxidizing conditions | Combined gas and inclusion defects |
| Poor cover flux control | Melt surface remains exposed | Variable gas content |
| Frequent re-melting of returns | Repeated exposure cycles | Process drift and cleanliness loss |
Does alloy chemistry change hydrogen behavior?
Yes, but the effect is not always simple. Alloying elements can influence:
- Surface oxidation tendency.
- Interaction with inclusions.
- Feeding characteristics during freezing.
- Effective porosity sensitivity.
- Response to degassing.
It is safer to say alloy chemistry modifies hydrogen-related defect risk rather than to claim one universal solubility change rule across all compositions.
General influence of alloy family
| Alloy family or feature | Typical hydrogen-related concern |
|---|---|
| High magnesium alloys | More reactive surfaces, more oxide and spinel formation |
| Silicon-containing casting alloys | Porosity may become prominent in critical sections |
| Recycled secondary alloys | Variable contamination and moisture history |
| High-quality billet alloys | Tight hydrogen limits due to downstream extrusion and surface demands |
| Rolling slab alloys | Blister and inclusion sensitivity during rolling and heat treatment |
Why does hydrogen porosity appear in some castings and not in others?
This is one of the most common plant questions. A melt can have moderate hydrogen content and still cast acceptably in one geometry, while another part fails badly. The explanation lies in the combined effect of hydrogen content, solidification rate, feeding ability, pressure, and oxide population.
Main conditions that influence whether pores actually form
- Dissolved hydrogen level before casting.
- Presence of pore nucleation sites such as oxides or bifilms.
- Solidification pressure in the mold or die.
- Cooling rate and mushy zone width.
- Ability of the casting to feed shrinkage.
- Local section thickness
- Mold design and gating turbulence.
Why high-pressure processes can mask part of the problem
Processes with high metal pressure may suppress visible gas pore expansion. That does not mean hydrogen is irrelevant. It may still affect internal quality, heat treatment response, or leak performance.
Defect expression by product type
| Product type | Common hydrogen-related defect |
|---|---|
| DC cast billet | Central porosity, extrusion surface variation, blister risk |
| Rolling slab | Internal gas defects, blisters after hot rolling or heat treatment |
| Gravity casting | Gas pores in thick sections, leak failure, poor machinability |
| Low-pressure casting | Internal porosity if melt quality slips |
| Die casting melt supply | Pressure tightness problems, trapped gas interacting with process defects |
| Wheel and structural casting | Reduced fatigue life and local shrinkage-gas interaction |
Why two identical molds can still give different results
If one ladle has been held longer, if one transfer path is more turbulent, or if one shift uses slightly different tool preheating, hydrogen and oxide levels can differ enough to change pore formation. This is why disciplined process control matters more than appearance-based judgment.
How do engineers and foundries measure hydrogen in molten aluminum?
Hydrogen cannot be controlled well if it is not measured. Leading plants combine direct and indirect methods depending on product value and required precision.
Common hydrogen and porosity evaluation methods
| Method | What it measures | Strength | Limitation |
|---|---|---|---|
| Reduced Pressure Test | Porosity tendency under vacuum | Fast and practical | Indirect, operator-dependent interpretation |
| Density Index | Relative gas tendency based on density difference | Useful trend control | Not a direct hydrogen concentration |
| First Bubble or similar analyzers | Direct dissolved hydrogen measurement | Strong process control value | Equipment cost and calibration discipline needed |
| Vacuum solidification sample | Porosity severity | Good comparative tool | Interpretation still needed |
| Metallography | Actual pore structure and inclusion interaction | Strong root-cause tool | Not a quick shop-floor method |
| Ultrasonic or X-ray on final product | Internal defect detection | Strong validation | Too late to prevent the melt problem |
Why density index remains so widely used
It is affordable, fast, and familiar. Plants can trend it heat by heat. The weakness is that it reflects porosity tendency, not only dissolved hydrogen. Inclusions and sampling practice can affect the result.
Why direct hydrogen analysis is valuable in premium operations
Premium billet, slab, and structural casting producers often prefer direct analyzers because they reduce ambiguity. Process development becomes easier when engineers can separate true hydrogen content from casting-sample variability.
Typical control strategy by plant maturity
| Plant level | Common practice |
|---|---|
| Basic foundry | Occasional reduced pressure test |
| Medium control operation | Routine density index plus operator logs |
| Advanced casthouse | Direct hydrogen analysis, trend charting, and statistical control |
| Premium aerospace or automotive supplier | Direct hydrogen measurement, inclusion control, recipe validation, and audit traceability |
Which methods reduce hydrogen content in molten aluminum most effectively?
The most effective method in modern production is rotary degassing using inert gas, usually argon or nitrogen, dispersed through a graphite rotor. Other methods can help, but rotary treatment remains the industrial standard in many plants.
Hydrogen control methods compared
| Method | Main mechanism | Strength | Limitation |
|---|---|---|---|
| Rotary degassing | Fine inert gas bubbles strip hydrogen from the melt | High efficiency, good repeatability | Needs equipment, graphite parts, and disciplined operation |
| Static lance bubbling | Gas bubbles rise through the melt | Simple setup | Lower efficiency due to coarse bubbles |
| Flux treatment | Helps inclusion removal and surface protection | Supports melt cleanliness | Limited hydrogen removal compared with rotary treatment |
| Vacuum treatment | Lowers hydrogen partial pressure | Strong in specific systems | Less common in many standard aluminum lines |
| Surface skimming only | Removes dross from top | Necessary housekeeping | Does not remove dissolved hydrogen |
| Better charge drying and low-turbulence handling | Prevents hydrogen pickup | Excellent preventive value | Cannot remove gas already dissolved |
Why prevention is cheaper than correction
The cheapest hydrogen is the hydrogen that never enters the melt. Dry charge, preheated tools, calm transfer, and covered holding practice reduce the burden on the degassing system.
Why rotary treatment also helps cleanliness
Fine gas bubbles generated by a graphite rotor can assist oxide flotation. This is one reason well-run degassing improves more than one defect category.
How does hydrogen control affect billet, slab, and foundry performance downstream?
Many teams think about hydrogen only in terms of pores visible on fracture surfaces. The true impact is broader.
Downstream effects linked to poor hydrogen control
- Surface blisters during rolling or reheating
- Lower pressure tightness in cast parts.
- Reduced fatigue life.
- Poor weld response in some products.
- Unstable machining.
- More scrap during ultrasonic or X-ray inspection.
- Customer complaints tied to leakage or visual quality.
- Inconsistent anodized appearance due to subsurface defects.
Why billets and slabs are highly sensitive
Wrought products undergo major deformation and later heat treatment. Hidden gas defects can open, enlarge, or become visible only after further processing. A billet that looks acceptable in the cast house may create downstream trouble in extrusion or rolling.
Product sensitivity table
| Product route | Why hydrogen control matters |
|---|---|
| Extrusion billet | Surface finish, blister resistance, profile consistency |
| Rolling slab | Sheet quality, foil integrity, blister control |
| Foundry castings | Leak tightness, machinability, mechanical properties |
| Automotive structural parts | Fatigue, crash behavior, internal integrity |
| Heat-treated castings | Pore expansion and reduced mechanical reliability |
What should engineers specify when writing a hydrogen-control standard?
A good process specification should not stop at “degas the melt.” It should define measurable acceptance criteria and operating conditions.
Practical items to include in a technical standard
| Specification item | Why it matters |
|---|---|
| Maximum allowable hydrogen content or density index | Sets a measurable quality target |
| Sampling frequency | Prevents “good heat, bad heat” drift |
| Melt temperature window | Solubility and treatment efficiency depend on temperature |
| Allowed holding time before casting | Limits reabsorption |
| Charge drying and tool preheat requirement | Reduces moisture-related pickup |
| Degassing gas type | Argon and nitrogen may behave differently in plant economics |
| Rotor speed, treatment time, and immersion depth | Makes the process repeatable |
| Criteria for graphite rotor replacement | Worn parts reduce bubble quality |
| Recordkeeping format | Supports root-cause analysis and audits |
Why acceptance limits should fit the product route
A gravity casting with modest pressure-tightness requirements does not need the same limit used in premium rolling slab. Process capability and customer need should shape the target.
Why trends matter more than single test values
One test result only gives a snapshot. A strong standard tracks trend by:
- Alloy
- furnace
- operator
- shift
- treatment recipe.
- downstream defect rate.
This reveals whether the root cause is stable or drifting.
What should procurement teams check when buying degassing equipment, graphite rotors, or hydrogen analyzers?
This article focuses on hydrogen solubility, yet many readers also need purchasing direction because the science directly drives equipment choices.
Procurement checklist for hydrogen control systems
| Item to evaluate | What buyers should ask |
|---|---|
| Rotary degassing unit | Can it maintain stable rpm, gas flow, and immersion depth? |
| Graphite rotor grade | What density, ash content, coating, and expected life are specified? |
| Graphite shaft design | Is alignment control strong enough to prevent wobble? |
| Gas delivery system | What purity and flow stability can be guaranteed? |
| Analyzer accuracy | What calibration routine and repeatability data are available? |
| Spare parts lead time | Can the supplier support urgent replacement needs? |
| Process support | Will the supplier help optimize treatment recipes in real production? |
| Documentation | Are operating procedures, drawings, and maintenance instructions included? |
Why total cost is more important than unit price
A low-cost rotor that produces unstable bubble size may increase:
- gas consumption
- treatment time
- scrap
- dross generation
- downtime
- customer risk
That means the cheapest spare is often the expensive one in real production.
How AdTech-related purchasing decisions connect back to solubility science
If liquid aluminum can hold much more hydrogen than solid aluminum, then every plant needs a way to control that gap before freezing begins. This is why degassing units, graphite rotors, analyzers, and melt-handling components are not optional accessories in demanding lines. They are direct responses to the solubility drop.
Which common misconceptions lead to repeated porosity problems?
Even experienced plants sometimes repeat the same mistaken assumptions.
Misconception 1: “The melt looked clean, so hydrogen must be low.”
A bright surface does not prove low dissolved gas. Hydrogen is invisible until it causes trouble.
Misconception 2: “Skimming removes hydrogen.”
Skimming removes dross and floating contaminants. It does not remove dissolved hydrogen from the bulk melt.
Misconception 3: “One degassing cycle fixes everything.”
Hydrogen can re-enter during holding, transfer, or turbulent pouring. Degassing must be combined with disciplined handling.
Misconception 4: “Only foundries need hydrogen control.”
Billet, slab, foil stock, and extrusion operations are highly sensitive too, often in ways that appear later downstream.
Misconception 5: “Porosity means only gas, not shrinkage.”
Many defects involve both gas and feeding limitations. Metallographic review usually shows the interaction.
Common mistakes and consequences
| Mistake | Immediate effect | Longer-term result |
|---|---|---|
| Charging wet returns | Rapid gas pickup | Persistent porosity across the shift |
| Degassing too early, then long holding | Reabsorption before casting | Good test, poor product |
| Overstirring the surface | Oxide entrainment | Combined gas and inclusion defects |
| Using worn graphite rotors | Coarse bubbles, weak degassing | Higher hydrogen and variable results |
| Ignoring analyzer calibration | False confidence | Poor process decisions |
How can plants reduce hydrogen pickup before the metal reaches the mold?
The strongest hydrogen control program begins before the melt enters the degassing station.
Preventive actions with high value
- Dry and preheat charge materials.
- Store returns under cover.
- Preheat tools, ladles, launders, and skimmers.
- Keep furnace atmosphere stable.
- Minimize open melt exposure time.
- Reduce turbulent transfer and waterfall pouring.
- Remove dross gently rather than stirring it into the bath.
- Verify gas purity and analyzer calibration.
- Replace worn graphite parts on schedule.
- Match treatment time to actual melt volume.
Why prevention and removal must work together
Prevention lowers the incoming hydrogen load. Degassing removes the remaining dissolved gas. Filtration then helps with suspended inclusions. The best metal quality comes from this layered strategy, not from relying on a single step.
What is the practical relationship between hydrogen solubility, degassing, and porosity risk?
This relationship can be summarized in one process chain:
- Moisture and exposure introduce hydrogen into molten aluminum.
- Liquid aluminum dissolves that hydrogen readily.
- Solid aluminum cannot hold the same amount.
- Solidification rejects hydrogen into remaining liquid.
- If hydrogen has nowhere to go, pores form.
- Porosity damages quality and increases scrap.
- Degassing and disciplined handling interrupt this chain.
Process chain summary table
| Step | Metallurgical event | Control method |
|---|---|---|
| Hydrogen pickup | Moisture interacts with liquid metal | Dry charge, preheating, controlled atmosphere |
| Dissolution in melt | Liquid aluminum holds hydrogen readily | Short holding time, covered melt handling |
| Solidification rejection | Solid rejects hydrogen into liquid | Lower starting hydrogen before casting |
| Pore nucleation | Hydrogen accumulates in last-freezing liquid | Degassing, low turbulence, clean melt |
| Final defect formation | Pores remain trapped in the product | Testing, process control, downstream inspection |
FAQs about hydrogen solubility in molten aluminum vs solid aluminum
Hydrogen in Molten Aluminum: 10/10 FAQ
1. Why is hydrogen more soluble in molten aluminum than in solid?
It comes down to atomic arrangement. Liquid aluminum has a disordered structure with more temporary “voids” and high atomic mobility, allowing hydrogen atoms to fit easily. Once it solidifies into a tight, face-centered cubic (FCC) crystal lattice, there is far less interstitial space available to hold the gas.
2. How much greater is hydrogen solubility in liquid aluminum?
The difference is dramatic. At the melting point, liquid aluminum can hold approximately 10 to 20 times more hydrogen than the solid phase. This sudden drop in solubility during the phase change is the physical root of almost all gas-related defects in aluminum casting.
3. What happens to hydrogen when aluminum solidifies?
As the metal freezes, the solid phase “rejects” the dissolved hydrogen into the remaining liquid. This causes the hydrogen concentration in the liquid to skyrocket until it reaches a point where it forms gas bubbles. If these bubbles are trapped by the advancing solid front, they remain as internal pores.
4. Is hydrogen the only gas of concern in aluminum?
5. Does higher melt temperature increase hydrogen solubility?
Yes. Solubility increases exponentially with temperature. “Overheating” the melt not only allows it to hold more hydrogen but also increases the rate at which the aluminum reacts with atmospheric moisture to pick up more gas.
6. Can skimming remove dissolved hydrogen?
7. What is the best industrial method to reduce hydrogen?
Rotary Degassing is the industry standard. By injecting fine bubbles of an inert gas (Argon or Nitrogen) through a spinning graphite rotor, the hydrogen atoms diffuse into the bubbles and are carried to the surface where they are safely released.
8. Why can a casting show porosity even after degassing?
9. How do plants test hydrogen in molten aluminum?
QUALITY CONTROL
The most common tool is the Reduced Pressure Test (RPT), which magnifies gas content for visual inspection. More precise operations use Density Index calculation or direct dissolved hydrogen analyzers (like the Alscan or Telegas) for real-time numerical data.
10. Why does hydrogen control matter in billet and slab production?
Final technical summary
Hydrogen solubility in molten aluminum versus solid aluminum is one of the most important concepts in aluminum metallurgy. The liquid metal can hold much more hydrogen than the solid metal. During freezing, that difference turns dissolved gas into a porosity threat. This is why melt treatment is not only about chemistry adjustment or surface skimming. It is about managing a thermodynamic transition that naturally pushes hydrogen out of solution at the worst possible time, right inside the product during solidification.
The practical lessons are clear:
- Liquid aluminum can absorb hydrogen easily, especially in hot, wet, turbulent conditions.
- Solid aluminum has very low hydrogen capacity.
- The solubility drop during freezing is the direct cause of gas porosity.
- Oxides, bifilms, and poor feeding make the defect more severe.
- Prevention begins with dry charge, dry tools, and calm melt handling.
- Control requires measurement, not visual guesswork.
- Rotary degassing and disciplined process control remain central in quality aluminum casting.
In AdTech process thinking, this topic is not a narrow laboratory issue. It affects equipment specification, melt handling design, degassing practice, graphite rotor selection, analyzer choice, and final customer quality. Any engineer, production manager, or procurement specialist working with aluminum should understand this solubility contrast clearly, because it explains why some heats cast cleanly while others generate porosity, blisters, and costly downstream failure.

