How does the deep bed filter influnces 3104 aluminum alloy part 1
1. Experiment
This test uses a Prefil-footprint slag measuring instrument to take samples at the outlet of the deep bed filter of the 3104 aluminum alloy melting and casting production line of an aluminum processing plant. The sampling conditions are shown in Table 1. The metallographic analysis and quantitative analysis of inclusions were carried out on the slag samples, and the types and quantities of inclusions in the aluminum alloy melt were obtained in different periods of use of the filter. Spectral analysis to get the type of inclusions filtered by the deep bed filter. During the casting process, it has a long service life, and the entire use process can be divided into early, middle, and late stages, and multiple castings can be performed in each stage. In the test, the use period A represents the first casting stage of the filter, and B, C, and D represent the early, middle, and late stages of the entire service life of the deep bed filtration, among which c1 and c2 are the mid-term use of the deep bed filtration. The start-up stage and the casting-end stage of the same casting.
2. Analysis of results
2.1 Analysis of inclusion types
At the outlet of the deep bed filter, the melt has good quality after a series of online treatments such as degassing and filtration, and the slag sample is relatively clean, with a small amount of inclusions at the interface between the filter cake and the melt. Judging from the inclusion morphology, the types of inclusions are fine oxide film, MgO, Cuboids (cuboid inclusions), spinel, and refiner residue TiB2 as shown in Figure 1. The fine oxide film, cuboid inclusions, and spinel are all oxides composed of A1, Mg, and O elements. The fine oxide film is in the shape of thin lines with branches, often with black plaque-like or granular inclusions. ; MgO is brown or black round fine granular inclusions; spinel (MgAl2O4.) is black granular or irregular-shaped inclusions, mostly from the reaction of MgO and Al2O3; cuboid inclusions are hard Black squares Oxides of Al, Mg, and O. These kinds of inclusions are similar in composition and often appear together.

Another common inclusion in the 3104 aluminum alloy melt at the outlet of it is the refiner residue TiB2. Due to the presence of B element, TiB2 was observed in the microscope as yellow or brownish yellow, and often aggregated into clusters with irregular shapes, as shown in Figure 1d. Due to the presence of inclusions, a small amount of TiB, particles adhere to the edge of the oxide film and the ceramic foam filter.
2.2 Quantitative analysis of inclusions
The metallographic observation of the Prefil sample was carried out, and the content of each inclusion in the slag sample was calculated by the area method, and the result was the area of inclusions per kilogram of aluminum melt (mm2/kg). Figure 2 shows the quantitative analysis results of inclusions in the melt at the outlet of it when the melt throughput is different.
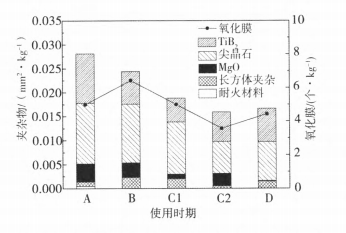
In the first casting stage of it, the retention capacity of inclusions is weak, and then with the increase of filtration tonnage, the number of inclusions in the melt gradually decreases, and the filtration efficiency increases. This phenomenon is related to the wettability between the aluminum melt and the alumina filter media. The wettability of aluminum melt to alumina is closely related to the melt purification process. Below 800℃, the bonding type of aluminum and aluminum oxide is a molecular bond; the wetting angle value is between150℃ ~160℃. Sarina Bao's study shows that the wetting angle 0 between aluminum melt and solid Al2O3, is exponentially related to time t, as shown in Eq. (1).
θ=A×exp(-t/t1) + θ0




